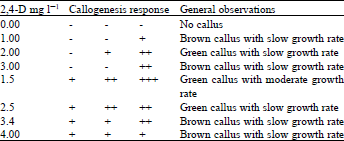
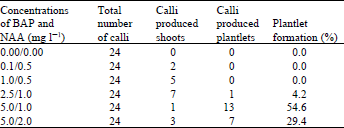
Research Article
Establishment of Tissue Culture Protocol in Brassica (B. napus L.)
Agricultural Biotechnology Programme, National Agricultural Research Center, Park Road, Islamabad, Pakistan
Hamid Rashid
Agricultural Biotechnology Programme, National Agricultural Research Center, Park Road, Islamabad, Pakistan
M. Ramzan Khan
Agricultural Biotechnology Programme, National Agricultural Research Center, Park Road, Islamabad, Pakistan