Research Article
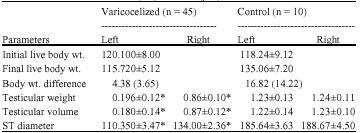
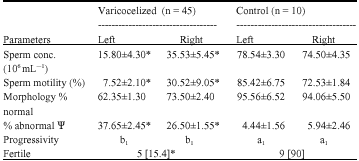
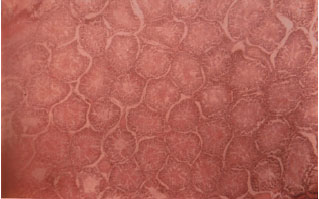
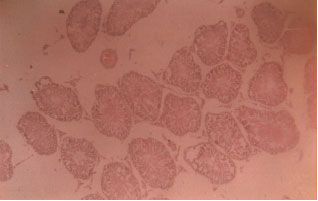
The Effect of Unilateral Varicocele on the Contralateral Testicular Histo-Morphology and Function in Rattus norvegicus
Department of Anatomy
P.I. Jewo
Department of Anatomy
I.O. Fadeyibi
Burns and Plastic Surgery Unit, Department of Surgery
S.O. Ikuerowo
Urology Unit, Department of Surgery, Lagos State University College of Medicine, Ikeja, Lagos, Nigeria