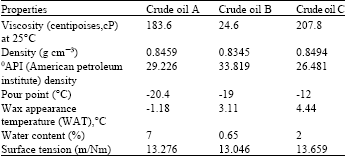
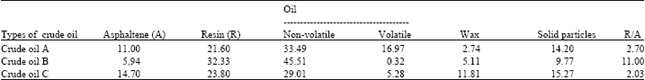
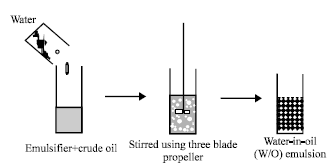
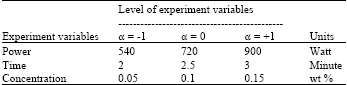Research Article
Destabilization of Heavy and Light Crude Oil Emulsions via Microwave Heating Technology: An Optimization Study
Faculty of Chemical Engineering and Natural Resources, Universiti Malaysia Pahang, Malaysia
Abdurahman H. Nour
Faculty of Chemical Engineering and Natural Resources, Universiti Malaysia Pahang, Malaysia
Azhari H. Nour
Faculty of Chemical Engineering and Natural Resources, Universiti Malaysia Pahang, Malaysia