Research Article
Impact of Storage Tanks on Drinking Water Quality in Al-Karak Province-Jordan
Department of Civil and Environmental Engineering, Faculty of Engineering, Mutah University, P.O. Box 67, 61710, Karak- Jordan
Due to water scarcity in Jordan, the development and regulation of the water distribution system for the public became eminent and mandatory to all areas in the country. Water rationing controlled the time schedule for the treated water supply especially in the summer months where water demand is the highest. Such adapted system encouraged the public to use domestic water storage tanks as water reservoirs for daily use. It is common to see such tanks over the roofs of residential houses, schools, hospitals and restaurants. The size of such tanks varies between one to three cubic meters; the majority of such tanks are made of galvanized steel. The domestic storage tanks are the main source of the water supply in addition to the network water distribution system when available. The domestic storage tanks are exposed indirectly to the surrounding environments such as wind, temperature, dust and others, which can impact the water quality of the storage tanks.
High concentrations of heavy metals could be significant if corrosion is evident in the tanks. Many studies over the past decades investigated the corrosion and controls of metals in water distribution systems[1-5]. The galvanized steel water storage tanks contain variable levels of heavy metals such as: Fe, Pb, Mn, Ni, Cu, Zn and many others. This contributes to the increasing concentration of these heavy metals in the drinking water. The affects of heavy metals on the quality of drinking water is very important as it becomes toxic to humans if exceeds certain concentration levels[6-9]. Researchers indicated positive correlations between high concentrations of trace metals in the drinking water and human health risk causing dangerous diseases such as cancer, sudden infant death and cardiovascular syndrome[10-12]. Many investigations were done on drinking water quality in Karak province. Jiries[13] investigated drinking water quality at source and found that anthropogenic activities were high at shallow water table levels. Khashman[14] indicated that dusty conditions had great impact on rainwater quality, which is the only source of groundwater recharge in Al-Krarak province.
The aim of this study was to assess the impact of storing water in galvanized steel storage tanks on the quality of drinking water from the physical and chemical prospectives under semiarid conditions such as Al-Karak Province in Jordan.
The study area: The investigated area is located in Al-Karak Province, 150 km south of Amman the capital of the country Jordan, which is the most populated area in the southern part of the country. The population is estimated to be over 220 thousand in 3495 km2 area[15].
The drinking water samples used in this investigation were obtained from three major towns in Al-Karak Province (Fig. 1) which are Mu’tah (Site I), Al-Marj (Site II) and Abu-Hamour (Site III), where the water sources are groundwater from Asultani, Aljoon and AL-quer Wells for Mutah, Ean Sara Springs, AL-quer Wells for Al-Marj and Al-quer Wells for Abu-Hamour, respectively.
The contact period between water and storage tanks is not more than three days, which is due to water rationing controlled program providing water to the investigated area twice a week. The tanks used in this study are made of galinized steel and many tanks were in wear and tear conditions. In some tanks, over 1 cm thickness of fine-grained sediments was observed at the bottom of the tanks.
Sampling and analysis: One hundred water samples during the summer of 2003 were collected from the residential galvanized steel water storage tanks from three different regions of Al-Karak Province. Pre-washed 300 mL polyethylene containers were used for sampling. Collection was duplicated at each site for quality assurance. One sample was used for major cations and anions, whereas the others were acidified with few drops of HNO3 for cations and heavy metal analysis. The water samples were kept refrigerated in polyethylene bottles and analyzed within 24 h after collection. A blank sample was made for each element determination in order to account for any analytical or instrumental error. Electrical conductivity and pH were determined immediately at sites when the samples were collected using a portable EC-meter model WTW LF320 and a portable pH-meter model WTW425.
Major anions and cations: Major Anions (F¯, Cl¯, NO3¯ and SO4-2) were analyzed by ion chromatography using Dionex-100 equipped with a conductivity detector model CDM-2 (sensitivity range; 0. 03-1000 μS cm-1) Analytical column AS4A-SC ion exchange, (250 * 4 mm ID) with an AG4A-SC guard column (50 * 4 mm ID) was also used for the separation of the major anions. Detection after suppression has been made with an ASRI anions self-regeneration suppressor. The injection volume was 25 μL and the flow rate was 2.0 mL min-1. The eluent was an aqueous solution consisted of 1.8 mM Na2CO3 and 1.7 mM NaHCO3. The chromatograms were registered on an Integrator 4400 Dionex.
Major cations (Na+, K+, Ca2+ and Mg2+) were analyzed using the same instrument with analytical column of CSI2 (250 * 4 mm ID), CG12 guard column (50 * 4 mm ID).
 | |
| Fig. 1: | Location map |
(CSRS-II cation self-regenerating suppressor and CDM-2 detector, from Dionex, USA). Analysis was carried out at 30 μS cm-1, with 1.0 mL min-1 of 20 mM methanesulfonic acid as eluent. The following concentrations of cations were detected at 2, 4, 8, 12 and 16 ppm Na+, K+, Mg+2 and Ca+2, respectively.
Heavy metals: Heavy metals (Pb, Fe, Cu, Zn, Mn and Ni) were measured by flame atomic absorption spectrophotometer model analyst 300 with standard graphite furnace model HAG 800 equipped with AS-72 auto-sampler. Data and run controlled were carried out using Winlab data management system.
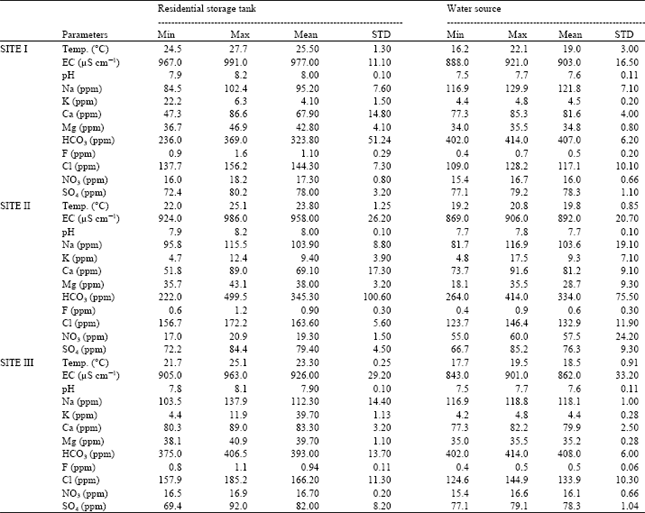
Statistical summary of the major ionic composition and heavy metals from the residential water storage tanks and water source for each site is given in Table 1 and 2 , respectively. The present results refer to the average of parameters collected and analyzed from thirty-five samples obtained from each of the three sites mentioned above. The results of both residential water storage tanks and its water source showed low variation of both ionic composition and heavy metal content. This could be attributed to the short period of contact between the tanks and input water where the water is renewed at least once every week.
| Table 1: | Maximum, Minimum and Mean±SD of major ions of storage water tanks and its source from different sites of Al-Karak province |
 | |
| Table 2: | Heavy metal concentrations in ppm for storage water tanks and its source water |
 | |
| ND: Not detected | |
The average electrical conductivity of the three sites showed low variation and ranged from 905 to 991 μS cm-1 (Table 1). However, all measured values in the residential water tanks were above the values of the water source for each site, which could be due to dust entering the storage tanks during the dusty summer season and also the elevated water temperature leading to further evaporation from the tanks. A similar result on increasing salinity in rainwater of the same area under dusty climatic conditions was reported by Khashman[14].
The ionic composition of water at source reflect the lithology of the area as the concentration of Ca, Mg and HCO3 make the highest contribution to the total mass of ions, comprising more than 48% of the total ionic species at all sites. The elevated concentrations of these ions reflect the carbonate lithology of the aquifers in the investigated area[16].
All samples showed elevated levels of Cl, which ranged from 137.7 to 185.2 ppm with an average concentration of 163.6 ppm. The high variation in Cl concentration is due to the different dosage of chlorination added at source.
Significant correlation coefficient was found between some ionic species indicating mineralogical dissolution from the aquifers. The significant correlation between (Ca+ Mg) with HCO3 (r =89.7) can be due to dissolution of carbonates (limestone and dolomite). On the other hand the high correlation between sodium and chloride (93.1) indicated the dissolution of halite in the ground water.
The existence of gypsum and calcite in the aquifers profile[13] provided a significant balance in the charge between ∑ (Ca2+ +Mg2+) to ∑ (SO42¯ and HCO3¯) of (98.5).
Nitrate level which is an indicator for contamination of pollution as it can result from the decomposition of nitrogenous organic compounds were relatively low (less than 21 ppm) at all sites where the water source was not from Ean Sara which contained elevated levels of nitrate ranging between 55 and 60 ppm. The source of such high nitrate values are in agreement with Salameh[16].
Although the residential storage tanks are made of alloy (Fe and Zn), the heavy metals at source were found in low concentrations which can be attributed to relatively high pH values enhancing precipitation and retard mobility of these metals if they exist in nature at source[17].
Despite the fact that the contact period between the water, network distribution system and the residential storage water tanks which are made mainly from iron and zinc is short (less than one week) elevated levels of these metals were found in the drinking water indicating dissolution of these metals to the water. This can be enhanced due to the fact that many storage tanks were rusty and not cleaned for a long period of time. Elevated levels of heavy metals in the drinking water from the residential storage tanks were noticeable in comparison to the water source, these levels were still below the drinking water standards in the country of Jordan[18] and didn’t exceed the World Health Organization (WHO) standards for drinking water[19].
The following can be concluded from this study:
| 1. | This research showed that the short contact period between the water and the galvanized residential storage tanks had no direct impact on the amount of heavy metals. |
| 2. | The water quality of stored water was similar to its source due to short storage time between the tanks and its water. |
| 3. | Heavy metals in storage tanks were higher than water at source, which is most probably due to the tanks wear and tear. |
| 4. | The quality of water at source was impacted by the dissolution of aquifer materials rather than anthropogenic origin. |
| 5. | The ionic concentration of all analyzed drinking water samples were below the local and international standard. |
The author would like to thank Professor Anwar Jiries for all his advice and support, Dr. Mufeed Batarsah for his help in the analytical procedures, Al-Karak Water Authority for the information provided and their support, also to the engineer and staff at the Wastewater Treatment plant/Mutah University and finally the student team who worked with dedication on this project.