Research Article
Study on Preparation and Some Properties of Panretin-Loaded Nanocapsules
Department of Engineering, Chemical Engineering Group, Islamic Azad University, Ghaemshahr Branch, Ghaemshahr, Iran
Panretin, or 9-cis-retinoic aicd or alitretinoin (Fig. 1) is an antineoplastic agent developed by Ligand Pharmaceuticals. It is a first generation retinoid. In the United States, topical alitretinoin (trade name Panretin) is indicated for the treatment of skin lesions in AIDS-related Kaposi's sarcoma. Panretin is not indicated when systemic therapy against Kaposi's sarcoma is required Under the trade name Toctino (marketed by Basilea, Switzerland) it has been granted prescription rights in the UK for oral use in chronic hand eczema. In May 2009 the National Institute for Health and Clinical Excellence (NICE) issued preliminary guidance on the use of Panretin for the treatment of severe chronic hand eczema in adults. The recommendation stated that only patients with severe chronic hand eczema who are unresponsive to potent topical corticosteroids, oral immunosuppressants or phototherapy should receive the drug.
Panretin is the lively form of a metabolic product of Vitamin A. It belongs to the cis-isomer of retinoic acid (Rigopoulos et al., 2004). This drug is triumphant in the topical behavior of different skin illness such as ichtiosys, acne vulgaris, psoriasis and neoplasias (Polano, 1974; Corbeil et al., 1994; Brisaert et al., 2001; Shin et al., 2005). The progress and dermal administration of topical systems included panretin in attendance some disadvantage for instance disadvantaged solubility, high chemical and photoinstability, which cause to be motionless metabolites and irritation of the treated region (Elbaum, 1988; Brisaert et al., 1995; Brisaert and Plaizier-Vercammen, 2000; Lin et al., 2000).
 | |
| Fig. 1: | Panretin, or 9-cis-retinoic aicd |
A lot of labors have been carried out over the last years to conquer some of these disadvantages. The relationship/addition of panretin with unlike type of release systems (niosomes, cyclodextrins, lipid nanoparticles) has enhanced its solubility (Montassier et al., 1997; Brisaert and Plaizier-Vercammen, 2000), chemical steadiness (Brisaert and Plaizier-Vercammen, 2000; Lin et al., 2000; Brisaert et al., 2001; Manconi et al., 2003; Lim et al., 2004; Ioele et al., 2005), bioaccessibility (Sacks et al., 1992; Ezpeleta et al., 1996; Lin et al., 2000; Shimizu et al., 2003; Shah, 2007). In excess of the past 10 years, polymeric nanocapsules have been widely considered as drug delivery service in the pharmaceutical meadow in addition to a well-organized coating material to control the drug discharge from microparticles (Beck et al., 2007). Nanocapsules are distincted as a matricidal polymeric arrangement, wherein drugs can be entrapped or molecularly detached. alternatively, nanocapsules are distinguished by a lipophilic center enclosed by a polymeric coating, wherein drugs can be suspended in the oil, detached inside the particle (Couvreur et al., 2002).
While these systems were handy epicutaneously, they can modify the drug pharmacokinetic and biocirculation through skin. In addition, their small sizes make feasible their formulation in dermatological products and facilitate easy use to the skin (Perugini et al., 2002; Guterres et al., 2007). Some researchers have been reported on the relationship of panretin in medium kind polymeric nanoparticles (Nam et al., 2003; Jeong et al., 2004; Seo et al., 2006). The endeavor of the current study was to get ready and typify panretin burdened nanocapsules equipped with the oil of sunflower seed-OSFS and to appraise their possible as topical release systems to get better the steadiness of panretin alongside photodegradation made by UV light.
This study was conducted from 2007 to 2009. Nanocapsule suspensions were organized (n = 3) by interfacial deposition of mentioned polymer technique (Fessi et al., 1988). temporarily, an organic solution created of panretin (0.0130 g), the oily phase, OSFS (0.81 mL), sorbitan monooleate (0.194 g), the polymer (poly-ε-caprolactone) (0.25 g) and acetone (68.0 mL) was added to an aqueous solution (134.0 mL) having polysorbate underneath reasonable magnetic stirring (20 min). afterward, the acetone was eradicated and the aqueous phase concentrated by evaporation underneath abridged pressure (40°C) to a ultimate volume of 25 mL (10 mg mL-1 of polymer and 0.60 mg mL-1 of drug). With the aim of confirm the influence of the polymeric layer, nanoemulsions were geared up omitting the polymer and blank formulations were organized omitting the drug. All planning were carried out secluded from the light and reserved in the dark throughout all the time. Drug content (mg mL-1) was strong-minded (n = 3) following dissolution of nanoemulsions in acetonitrile (1 mL of suspension to 25 mL of acetonitrile) and examined by high performance liquid chromatography (HPLC). The chromatographic system consisted of a Gemini RP-18 column (150x4.60 mm, 5 μm, Phenomenex, Torrance, USA) and a Shimadzu instrument (LC-10AVP Pump, UV-vis SPD-10AVP Module, Class-VP Software, Shimadzu, Tokyo, Japan). The movable phase at a flow rate of 1.0 mL min-1 be composed of acetonitrile/ water (85:15%, v/v) containing 1% of glacial acetic acid. The volume introduced was 20 μL and panretin was alleged at 340 nm. The technique was linear (R2 = 0.9999) in the range of 1-20 μg mL-1. The specificity was tested in attendance of the colloidal suspension adjuvant and demonstrated that these factors did not change the panretin assay. The percent of encapsulation efficiency (%) was planned by the variation among the whole and free drug concentrations. The pH values of the suspensions were determined and the particle sizes and polydispersity indices (n = 3) were calculated by photon correlation spectroscopy (3 measures/batch; 2 runs of 30 sec/ measure at 25°C) following sufficient dilution of an aliquot of the suspension in water. The zeta potential values were determined (3 measures/batch; 10 runs/measure at 25°C) after dilution (1:500) of the suspensions in 1 mM NaCl (Zetasizer Nanoseries, Malvern Instruments, Worcestershire, UK). All formulations appeared macroscopically homogeneous and their aspects were alike to a milky bluish opalescent fluid (Tyndall effect), despite of the kind of oily phase (OSFS) or the vesicle construction (nanocapsule or nanoemulsion).
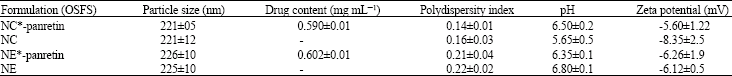
The physicochemical characteristics of the formulations are accessible in Table 1, as shown in Table 1, The formulations presented drug content (mg mL-1) on the edge of their theoretical value (0.6 mg mL-1) with towering encapsulation efficiency (>99.9% for all drug-loaded formulations). Exact amounts of the panretin associated in nanocapsules could not be assayed because its concentration in ultrafiltrate was lower than the quantification limit (0.06 μg mL-1) of the HPLC method. Panretin -loaded NC and NE presented similar mean diameters (220-226 nm), acidic pH values (5.1-6.7), negative zeta potentials (between -4.4 and -10.8 mV) as well as polydispersity indices below 0.25 indicating an adequate homogeneity of these systems which is in supported with Alves et al. (2007). The photodegradation of panretin was calculated using an UV artificial lamp (Phillips TUV lamp-UVC long life, 40 W). The panretin solution or panretin loaded nanoparticle formulations (2 mL in a 1 cm quartz cuvette perfectly stoppered) were exposed to UV radiation for 2 h at an unchanging distance of 10 cm (n = 3). With the purpose of quantify the panretin by HPLC, Hundred microliters of the samples were reserved every 10 min and diluted with acetonitrile. In order to refute the theory of thermal degradation and to be protected from UV light, panretin solution and nanoparticle formulations enclosed by aluminum foil were moreover evaluated in the similar method. The photodegradation profile of panretin solution (36.7±2.2%) of the first drug concentration remained after 1 h) was along with a first order kinetic, whereas the data attained from the photodegradation experiments of the panretin-loaded nanocapsule or nanoemulsion fitted improved to the zero order kinetic.
| Table 1: | Physicochemical characteristics of drug-loaded and drug-unloaded colloidal systems prepared with OSFS Formulation |
 | |
| Table 2: | Half-time (t1/2) and n rate constants rate of photodegradation (k) of free panretin and panretin loaded nanocapsules or nanoemulsion exposed to UV light for 1 h (n = 3) |
 | |
When panretin in solution or loaded in formulations were secluded from light, more than 90% of panretin stay behind intact after 1 h of irradiation. The half-lives (t1/2) and photodegradation rate constants (k) of free and panretin-loaded nanoparticles bared to UV light for 1 h are shown in Table 2. Panretin solution showed a half-life time around 40 min, while the panretin-loaded nanocapsule suspensions showed half-life times between 77 and 97 min. As it is noticed in Table 2, the nanoemulsions in addition presented a potential to improve the photostability of panretin. Nevertheless, this enhancement was lower t1/2 involving 56 and 84 min compared to the nanocapsules t1/2 between 77 and 97 min. These consequences demonstrate the significance given by the attendance of the polymer to put off the photodegradation of panretin. This enhanced fortification presented by the nanocapsules alongside UV-induced photodegradation of alitretinoin could be accredited owing to the crystallinity of the polymer, which has the aptitude of reflecting and dispersion UV radiation which is adapted by Perugini et al. (2002) and Jimenez et al. (2004). Alike protection was earlier reported for other nanoparticulate systems similar to liposomes (Ioele et al., 2005), niosomes (Manconi et al., 2003) and complexes with cyclodextrins (Lin et al., 2000). On the topic of the sort of oily phase, the employ of unlike oily phases did not show any arithmetical dissimilarity on the guard alongside photodegradation.
The current study demonstrated for the first time the viability to get ready panretin-loaded nanocapsules, at a concentration of 0.6 mg mL-1, by means of oily phase (OSFS). Panretin-loaded nanocapsules enhanced panretin photostability and symbolize a potential system to be included in new topical or systemic dosage forms containing panretin.