ABSTRACT
Effects of Al and Ca interactions on the sugar compositions and molecular mass of hemicellulosic polysaccharides were examined in the roots of an Al sensitive wheat cultivar (Triticum aestivum L.). Seedlings were grown in nutrient solution for 7 days and then exposed to treatment solutions containing Al (0, 50 μM) and Ca (0, 2500 μM) in a 500 μM CaCl2 solution at pH 4.5 for 8 days. High Ca supply (2500 μM) improved the root growth significantly. The contents of pectin and hemicellulose were increased under Al stressed condition and this increase was conspicuous in hemicellulosic polysaccharides. The increase in the hemicellulose content was attributed to increases in arabinose, xylose and glucose in neutral sugars. Calcium supply decreased a little these contents in Al stressed cell walls. The elution profile of molecular mass of hemicellulosic polysaccharides was shifted to higher molecular mass region by Al stress compared to control and the Ca treatment with Al shifted slightly lower molecular mass regions. Aluminum treatment increased the estimated arabinoxylan and β-glucan contents and Ca treatment with Al decreased these contents. Aluminum treatment decreased significantly the β-glucanase activity, whereas Ca treatment with Al increased the activity. These results suggest that the modulation of the activity of β-glucanase with Al stress may be involved in part in the alteration of the molecular mass of hemicellulosic polysaccharides. The increase in molecular mass of hemicellulosic polysaccharides with Al stress may induce the mechanical rigidity of the cell wall and inhibit the root growth. High Ca treatment with Al is considered to reduce the mechanical rigidity of the cell wall and improve root growth.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajps.2005.11.16
URL: https://scialert.net/abstract/?doi=ajps.2005.11.16
INTRODUCTION
Aluminum toxicity and differential Al tolerance in plants have been the subjects of intensive investigation in plant nutrition. Despite the efforts of many researchers, little is known about the contribution of non-metabolic factors such as cell wall components to Al tolerance mechanisms. Since accumulation of Al in the cell walls has been suggested as a possible tolerance mechanism[1], differences in cell wall chemistry influence these mechanisms. Aluminum strongly binds to the cell wall of root epidermal and cortical cells[2]. The Al bound to the cell wall plays an important role in the inhibition of cell elongation[3]. Biochemical modifications of the cell wall, such as changes in the molecular size and quantities of cell wall polysaccharides, have been considered to be involved in the regulation of cell wall extensibility[4,5]. Alteration of the molecular mass of cell wall polysaccharides and mechanical extensibility of cell wall in response to various environmental stress have been reported in various plant materials[6]. Increases in the amount of polysaccharides causing the thickening of cell wall may be one of the mechanisms by which Al decreases cell wall extensibility[7]. The chemical and mechanical properties of the cell wall seem to be modified by Al because Al binds strongly to the cell wall of roots thus causing growth inhibition[7]. However, changes in the mechanical properties of cell wall by Al are still speculative in wheat roots.
The interaction between Al and Ca is an important factor affecting Ca uptake and transport in plants, Al injury and plant growth[8]. Al ions are positively charged under acidic condition in the forms of Al3+, Al(OH)2+ and Al(OH)2+. Therefore, negatively charged sites are the preferential sites for Al sorption. Cell walls contain highly negatively charged sites where Al3+ and Ca2+ perhaps compete with each other. There is a report that Al ions have much higher preferences over bivalent Ca2+, which enable them to replace of Ca2+ in the cell wall[9]. So, there must be interaction effects between Al and Ca in the plant cell walls. In the presence of rhizotoxic levels of Al, supplementation of the medium with higher level of Ca2+ alleviates growth inhibition[10]. Several mechanisms for Ca2+ alleviation of mineral toxicity have been proposed. A commonly proposed one is the restoration of toxicant-displaced Ca2+[11,12]. However, how Ca affects the hemicellulosic polysaccharides under Al stress conditions still remains unknown.
The present study deals with the interaction effects of Al and Ca on the modification of cell wall hemicellulosic polysaccharides in wheat plants.
MATERIALS AND METHODS
Root growth: The seeds of Kalyansona which had been evaluated as an Al sensitive cultivar[13] were surface-sterilized with 5% sodium hypochlorite and stirred for 10 min to eliminate pest contamination. The treated seeds were washed and soaked with distilled water and kept in refrigerator for 24 h. Then the seeds were sown in a sieve plate floating on full nutrient solution in a large plastic container at pH 5.0. After seven days, wheat seedlings with similar root length were placed on slide mounts with nets (4 plants/mount) and pre-cultured for 7 days in 3.5 L plastic container with full nutrient solution at pH 5.0. The solution was continuously aerated with air pump. The full nutrient solution was composed of N (50 mg kg-1) as NH4NO3; P (20 mg kg-1) as Na2H2PO4·2H2O; K (40 mg kg-1) as K2SO4; Ca (20 mg kg-1) as CaCl2·2H2O; Mg (24 mg kg-1) as MgSO4·7H2O; Fe (2 mg kg-1) as Fe-EDTA; B (0.5 mg kg-1) as H3BO3; Mn (0.5 mg kg-1) as MnSO4·5H2O; Zn (0.2 mg kg-1) as ZnSO4·7H2O; Cu (0.05 mg kg-1) as CuSO4·5H2O; Mo (0.05 mg kg-1) as (NH4) 6Mo7O4·2H20. Pre-culture solutions were replaced by treatment solutions containing Al (0, 50 μM) and Ca (0, 2500 μM) in a 500 μM CaCl2 solution at pH 4.5 with three replications. Treatment solutions were renewed every day with fresh solutions and pH was monitored at 4.5 using diluted HCl and NaOH. At 8 days after treatment the plants were harvested and the root length and fresh weight were measured. A small portion of roots was dried at 70°C for 48 h for measuring Ca and Al contents of roots. Aluminum content of roots was determined by calorimetric method using pyrocatechol violet and hexamethylene tetramine buffer solution[14] after digesting plant samples with concentrated H2SO4 and H2O2. Calcium content of roots was determined by atomic absorption spectrophotometry. The results were expressed as mean values of measurements with replications±standard deviation (SD). The remaining all fresh root samples were instantly frozen in liquid nitrogen and stored at -80°C in deep freezer for collecting the CWM.
Collection of CWM: The cell wall components were extracted according to Dawson et al.[15] with slight modifications. The root samples were grinded to a fine powder in a warring grinder pre-cooled with liquid nitrogen. The powder was then homogenized in a homogenizer with PAW (2:1:1 w/v/v) until the tissue thawed and became fine slurry. The slurry was centrifuged at 5000 g for 20 min at 4°C and the pellet was further washed with PAW solutions followed by two washes with cold distilled water and centrifuged. Finally the insoluble material was treated for 24 h with α-amylase in 0.1 M Mops (pH 7.0) for degrading starch and the insoluble pellet was dialyzed against distilled water and lyophilized to give the CWM.
Fractionation of CWM: Sequential extraction of the CWM was undertaken as described by Selvendren and Ryden[16], with slight modification. The CWM (750 mg) was suspended in 75 mL of 0.05 M acetate buffer (pH 6.5) containing 0.05 M CDTA and stirred for 16 h at 25°C. The suspension was centrifuged (5000 g for 20 min at 4°C) and the pellet was washed (stirred about 4 h) with 75 mL cold distilled water and centrifuged. The combined supernatants were dialyzed exhaustively against distilled water and lyophilized to give the CDTA soluble pectin fraction.
To the CDTA insoluble pellet 75 mL of 0.05 M Na2CO3 containing 0.02 M NaBH4 was added and the suspension was incubated with constant stirring for 16 h at 25°C. The suspension was centrifuged (5000 g for 20 min at 4°C) and the pellet washed twice with 75 mL of cold distilled water and centrifuged. All supernatants were combined and dialyzed exhaustively against distilled water and lyophilized to give the Na2CO3 soluble pectin fraction.
The Na2CO3 insoluble CWM was extracted with 4 M KOH containing 20 mM NaBH4 and stirred for 24 h under nitrogen gas at room temperature. The supernatants were recovered by centrifugation, cooled in an ice bath, neutralized with glacial acetic acid, recovered by exhaustive dialysis for 48 h and lyophilized to give the alkaline soluble hemicellulosic fraction. The aliquot (5 mg) KOH soluble hemicellulosic fraction was hydrolyzed with H2SO4 at 100°C for 16 h.
Gas liquid chromatography: The neutral sugar composition of KOH soluble hemicellulosic fraction was analyzed by GLC. Acetylating of the sugars after conversion to alditol acetate was performed according to Blakeney et al.[17] GLC was carried out on a Shimadju GC-18A apparatus equipped with a flame ionization detector. A capillary column (25 m, 0.22 mm i.d., 0.25 μm Hicap CPB10) was used and operated at 220°C with gas flow rate of 60 mL/min of nitrogen. Peak areas were measured with a Shimadzu Chromatocorder-21. The content of neutral sugars was calculated from the GLC peak area ratio.
Total sugar and uranic acid analysis: Total sugar content of hemicellulosic fraction was quantified by the phenol sulphuric acid method[18] using glucose as standard. Uranic acids were measured by modified carbazole-sulphuric acid method[19] using galacturonic acid as standard.
Determination of the molecular mass of hemicellulosic polysaccharides: Gel chromatography of the lyophilized hemicellulosic fractions in each treatment was carried out on a Sepharose CL-6B column (90x2.5 cm) which had been equilibrated with 0.05 M phosphate buffer (pH 7.8). The column had been calibrated with authentic dextrans (10, 70, 120 and 510) purchased from Pharmacia and Sigma Chem. Co. The samples (ca.5 mg) were dissolved 5 mL of the 0.05 M phosphate buffer (pH 7.8) and eluted with the same buffer at a flow rate of 20 mL h-1. The elute was collected in 5 mL fractions and analyzed for total sugar as described above.
β-glucanase activity: The roots were excised and were immediately frozen with liquid nitrogen and kept at -80°C until use. Extraction and assay of β-glucanase activity carried out essentially by the methods of Chen et al.[20]. The frozen roots (ca. 400 mg in fresh weight)were homogenized with ice cold 10 mM sodium phosphate buffer (pH 7.0).The homogenate was filtered through propylene mesh (32 μm). The fraction was washed with the same buffer and then suspended in 10 mM sodium phosphate buffer (pH 6.0) containing 1M NaCl. The suspension was kept for 24 h at 4°C and filtered through propylene mesh. The filtrate was used as enzyme extract for the measurement ofβ-glucanase activity. The reaction mixture (total 100 μL) contained 50 μg of β-glucans (β-1, 3; 1, 4-D-glucans) and 5 μg of cell wall protein in 10 mM sodium phosphate buffer (pH 6.0).The mixture of solution was incubated for 6 h at 37°C. After the incubation the reaction was terminated by boiling. Enzyme activity was determined by monitoring the increase in reducing sugars liberated from β-glucans by the Somogyi-Nelson method[21] and expressed as glucose equivalent. Protein content was determined with a protein Assay Kit (Bio-Rad, Herculus, CA, USA).
RESULTS
Root growth: Interaction effects of Al and Ca are shown in Fig. 1. The 2500 μM Ca treatment improved root length by 31% under Al stressed conditions (Fig. 1). The Al treatment decreased Ca content of roots and Al content was decreased by the Ca treatment significantly (Table 1).
Cell wall polysaccharides: The Al treatment increased the contents of pectin and hemicellulose in the cell wall of roots. The Ca treatment with Al treatment caused a reduction of the contents of pectin and hemicellulose (Table 2).
Sugar compositions: In the hemicellulosic fraction, the neutral sugar part was composed of mainly xylose and arabinose and others are glucose, fucose, galactose, rhamnose and mannose. The Al treatment increased greatly the contents of arabinose, xylose and glucose in comparison with those without Al treatment (Table 3). The Ca treatment decreased slightly the contents of arabinose, xylose and glucose relative to the control in the absence of Al and in the presence of Al it decreased greatly the amount of glucose, arabinose and xylose (Table 3).
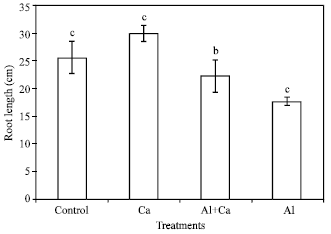 | |
| Fig. 1: | Effects of Al and Ca interactions on root length of wheat plants. Means with different letter are significantly different (p≤0.05, Duncan New Multiple Range Test); vertical bars indicate the SD of means of three replications |
| Table 1: | Mean±SD of Al and Ca interactions on Al and Ca content of roots of wheat plants |
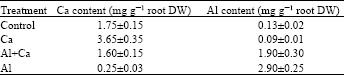 | |
| Table 2: | Effects of Al and Ca interactions on the contents of pectin and hemicellulose in the cell wall of wheat roots. Means with different letters are significantly different (p≤0.05, Values are means±SD (n=3) |
 | |
The arabinoxylan content was calculated as the combined amount of arabinose and xylose present in the CWM. The arabinoxylan content was higher in the Al treatment than in the control and Ca treatments. The Al treatment significantly increased the arabinoxylan content. The Ca treatment with Al treatment slightly decreased the arabinoxylan content relative to the Al treatment alone (Table 3). The glucose content was conspicuously increased by the Al treatment and the Ca treatment decreased it a little in the presence of Al.
Molecular mass of hemicellulosic polysaccharides: The molecular mass distribution of hemicellulosic polysaccharides in the cell wall was determined by phenol-sulfuric acid method on gel permeation chromatography (Fig. 2). Hemicellulosic polysaccharides were eluted to higher molecular mass regions with Al-stress compared to those without Al stress. Calcium treatment with Al shifted the elution to a little lower molecular mass regions.
β-glucanase activity: Table 4 shows β-glucanase activity in a protein fraction extracted with 1M NaCl from the cell walls. The activity of β-glucanase per unit of protein content (per μg protein) was decreased significantly with Al-stress and the activity was increased by high Ca treatment with Al-stress (Table 4).
DISCUSSION
Significant improvement of Al injury in wheat plants was observed with the addition of high Ca under Al stress conditions (Fig. 1). The Ca treatment decreased the Al content of roots (Table 1) and caused a substantial change in the chemical properties in the cell wall (Table 3). Several hypotheses have been proposed to explain amelioration of Al toxicity by Ca in plants. For example, Ca alleviates Al toxicity by reducing Al3+ activity at the surface of plasma membrane and thus decrease the Al uptake by roots[22], increasing ionic strength of the solutions[23], competing with Al ions to bind at sensitive sites in either symplast or apoplast of roots[24] and enhancing internal detoxification of Al.
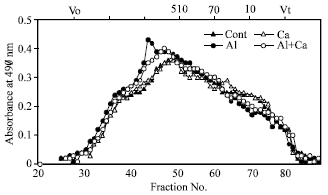 | |
| Fig. 2: | Effects of Al and Ca interactions on elution profiles of hemicellulosic polysaccharides of roots in wheat plants. Vertical bars denote the elution position of molecular mass standards (kDa) and void volume (Vo). Each elution profile is means of three replications without SE |
On the other hand Al treatment reduced the Ca content of roots (Table 1). Aluminum might exclude some part of the Ca from root exchange sites, which must be occupied by Ca for normal root elongation. Considering all the reports so far, high Ca supply can improve obviously root growth under Al stress conditions. Furthermore, Ca influences the compositions of cell wall polysaccharides. The result of increase in the hemicellulose content in the cell wall under Al stressed conditions (Table 1) is consistent with the reports on squash roots by Le Van et al.[25] and wheat roots by Tabuchi and Matsumoto[7]. This increase may be related to the rigidity of cell wall. The rigidity is considered to be one of the mechanisms by which plant roots can survive against Al toxicity. High Ca supply might modify the metabolism and synthesis of hemicellulose under Al stress conditions.
Aluminum treatment increased molecular mass of hemicellulosic polysaccharides in the cell wall of roots (Fig. 2). The molecular mass is related to the viscous state of cell wall and involve in regulation of the mechanical extensibility of cell walls[5]. A reduction in molecular mass of hemicellulosic polysaccharides in high Ca treatment with Al-stress (Fig. 2) may contribute to increase the mechanical extensibility of the cell wall.
The neutral sugar composition of hemicellulose fraction (Table 3) indicates that xylose, arabinose and glucose are the main sugars assuming that they are derived from arabinoxylan and β-glucan which are the main hemicellulosic polysaccharides of cereals[26,27].
| Table 3: | Effects of Al and Ca interactions on the content of neutral sugars in the hemicellulose of cell wall of wheat roots. Rha, rhamnose; Fuc, fucose; Ara, arabinose; Xyl, xylose; Man, mannose; Gal, galactose; Glc, glucose |
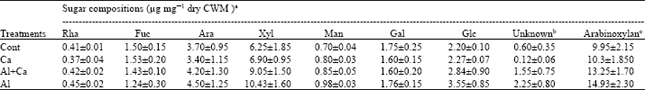 | |
| aEach value is the mean of three replications ±SD, bIn each treatment, the identifies of some unknown components were not confirmed cArabinoxylan was calculated as the combined amount of arabinose and xylose | |
| Table 4: | Effect of Al and Ca interactions on β-glucanase activity in roots of wheat plants. Means with different letter are significantly different (p>0.05). Values are means±SD (n=3) |
 | |
Some xylose is also derived from xyloglucan and some arabinose from glycoprotein but xyloglucan and glycoprotein contents are very trace in monocotyledonous[28,29]. So, the total amount of arabinose and xylose (Table 4) can be considered to be the content of arabinoxylan in the cell wall. A higher glucose content with Al-stress may be originated from the higher content of callose (β-1, 3; 1, 4-D-glucans) in the cell wall. These ideas suggest that the high molecular mass of hemicellulosic polysaccharides in Al treatment (Fig. 2) may be related to the increases in arabinoxylan and β-glucan synthesis in the cell wall. The results of β-glucanases activity clearly showed that Al treatment decreased the activity in the cell wall (Table 4). The decrease in the activity may involve at least in part in the increases of molecular mass of hemicellulosic polysaccharides with Al-stress because β-glucan are eluted in high molecular mass regions[30]. Therefore, the obvious changes in molecular mass of hemicellulosic polysaccharides (Fig. 2) suggest the possibility that Al treatment affects the metabolic turnover of the β-glucans. The decrease in estimated β-glucan by Ca treatment with Al stress indicates that the synthesis of this substance is normalized by influencing the β-glucanase activity and the molecular mass of hemicellulosic polysaccharides is reduced (Fig. 2) and reduce the rigidity of cell wall.
It can be concluded that Al treatment increased the mechanical rigidity of the cell wall by increasing the matrix polysaccharides in the cell wall. The increased molecular mass of hemicellulosic polysaccharides by Al may reduce the mechanical extensibility of the cell wall and inhibit root growth. High Ca treatment with Al may maintain normal synthesis of the matrix polysaccharides even under Al-stress. However, the further work is needed to estimate the absolute contents of matrix polysaccharides i.e arabinoxylan and β-glucan in the cell wall of wheat roots and the activity of several enzymes related to the synthesis of arabinoxylan with Al stress to confirm the inferences of this study.
REFERENCES
- Delhaize, E., P.R. Ryan and P.J. Randall, 1993. Aluminum tolerance in wheat (Triticum aestivum L.) (II. Aluminum-stimulated excretion of malic acid from root apices). Plant Physiol., 103: 695-702.
Direct Link - DuBois, M., K.A. Gilles, J.K. Hamilton, P.A. Rebers and F. Smith, 1956. Colorimetric method for determination of sugars and related substances. Anal. Chem., 28: 350-356.
CrossRefDirect Link - Blakeney, A.B., P.J. Harris, R.J. Henry and B.A. Stone, 1983. A simple and rapid preparation of alditol acetates for monosaccharide analysis. Carbohydr. Res., 113: 291-299.
CrossRefDirect Link








