ABSTRACT
Plant breeders are much concerned about the durability of the resistance gene(s) that they are incorporating into the better performing backgrounds to tolerate biotic stresses. While resistance that is controlled by a single major gene (vertical resistance) is simple to achieve, the one controlled by poly genes (horizontal resistance) is not. There are a number of ways to make the non-durable resistance genes durable. Comprehensively, the exploitation of genes with known durability, the use of quantitative resistance and the use of gene deployment are suggested to be some of them that can lead to the better ways of achieving durability of resistance. There are also various approaches of deploying genes to achieve durability of resistance. Therefore, to realize durability of disease resistance genes in crop plants any one of the approaches that are dealt in this study can be used without compromising their economic feasibility.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajps.2004.618.623
URL: https://scialert.net/abstract/?doi=ajps.2004.618.623
INTRODUCTION
The contribution of crop improvement to feed the ever-growing world population is quite eminent. However, it also generates some undesirable effects like genetic erosion and vulnerability of the improved cultivars to diseases and other biotic and abiotic stresses. During the co-evolution of plants and pathogens, all the important crops are damaged unless effective protection is devised. Protection may be realized by different means: chemicals, hygiene, biological control or host plant resistance[1]. Host plant resistance is the most preferable means of crop protection of all kinds as it combines the advantages of cost effectiveness and ecological soundness. However, it has been a challenge to the breeders to achieve durable resistance. Still there have been some degree of success in achieving durable resistance even though we still have not fully understood what actually causes a resistance to be durable, nor how to distinguish durable resistance from non durable resistance, nor even what criteria to use to decide whether a resistance is durable[2]. The exploitation of genes with known durability, the use of quantitative resistance and the use of gene deployment can lead to the better ways of achieving durability of resistance. This study intends to review the various approaches of gene deployment to ensure durability of resistance to diseases.
Genetic vulnerability
The milestones: The consequences of genetic vulnerability are so devastating that it would be mandatory to remind some of the events that occurred in the history of global agriculture. The Irish famine in Ireland, England and continental Europe in 1840s that occurred due to Potato Late blight (Phytophthora infestans) and took millions of lives; the wheatless days in the USA in 1917 that wiped out the wheat fields occurred by wheat stem rust (Puccinia graminis); the complete elimination of all oats derived from the variety Victoria in the USA in the mid 1940s by Victoria blight (Cochliobolus victoriae); the destruction of maize hybrids carrying T-cytoplasms in the USA in 1970-71 by Southern corn leaf blight (C. heterostrophus) and the great Bengal famine in India in 1943 due to the destruction of rice by C. miyabeanus are some of them to remember.
What causes genetic vulnerability?: It is obvious that crop improvement has given the best cultivars of the agriculturally important crop species, which are rewarding the farmer with their yields, the trader and the consumer with their qualities. On the contrary, the widely cultivated improved varieties are genetically uniform, which invites them to a risk of disease epidemics. Genetic vulnerability, the susceptibility of most of the cultivated varieties of a crop species to a disease, insect pest, or some other stress due to similarities in their genotypes, usually due to the presence of one (generally more) common parent(s) in their ancestry[3], can be caused by: a) Narrow genetic base: Many of the improved varieties have one or more parents in common with each other; b) devoting a large acreage of land for the cultivation of a single cultivar will facilitate disease epidemics. Because of the wide use of genetically uniform varieties a situation known as the ‘boom’ and the ‘bust’ cycle occurs. This concept was first proposed by Priestley[4] and it describes the situation, particularly with regard to cereal varieties. When a new variety with a vertical resistance is developed it soon becomes popular and the area under this cultivar increases. This stage is called as the boom. However, when a new race of the pathogen starts to appear and epidemic occurs, the cultivar loses its popularity and the area under it declines. This stage is known as bust (Fig. 1). There are various examples of these sorts[5,6]. c) Introduced pathogens: This is exemplified by the late blight epidemic on potatoes in Europe that took millions of lives and responsible for the migration of a large number of people to the neighboring countries; d) Induced susceptibility with desirable traits: there are again good examples for this. One very good example is the destruction of maize hybrids carrying T-cytoplasms in the USA[7]. Another example is the victoria blight epidemic; e) the otherwise resistant varieties can be attacked by the previously unrecognized strains of pathogens; f) Failure of vertical resistance: vertical resistance is governed by one or a few major genes, which are of no durability. The break down of vertical resistance is a common phenomenon in breeding for resistance; g) Environmental factors such as increased host plant density, use of plenty of fertilizers and irrigation.
Disease resistance: It is the inherent ability of a plant to prevent or restrict the establishment and subsequent activities of a potential pathogen[8]. Resistance is of two types viz., vertical resistance and horizontal resistance[9]. According to Vanderplank[10] vertical resistance is effective only against the initial inoculum reaching a field or a plot. Only horizontal resistance remains in the second cycle onwards with some exceptions.
Durability of resistance: Resistance that remains effective in a cultivar that is widely grown for a long period of time in an environment favorable to the disease is said to be durable resistance[11]. It is exemplified by the “field resistance” of potatoes to the late blight disease[12].
Major gene resistance against biotrophic pathogens such as mildew and rust is generally highly unstable and non-durable. Polygenic resistance on the whole appears to be durable[13]. In cases, where major genes are involved it is usually not difficult, but when the resistance is of a quantitative nature various problems may arise in trying to distinguish the more resistant entries from the less resistant ones[14]. Although the pathway to durable resistance is very narrow, there have been some outstanding successes.
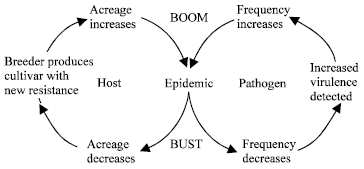 | |
| Fig. 1: | The boom and bust cycle[4] |
According to Parry[13] a winter wheat variety Cappele-Desprez, which occupied more than 80% of the total wheat area in Britain for 10 years and was a widely grown variety for over 20 years, was adequately resistant to both yellow rust and eyespot caused by Pseudocercosporella herpotrichoides.
Mechanisms of durable resistance: Resistance conferred with major genes has frequently failed to provide long-term disease control and the use of single major genes in cultivars grown over a broad area potentially lead to serious epidemics[15]. It is emphasized that durability of resistance is not merely a property of genes; it is the property of the cropping system. The strategies by which durability of resistance can be achieved include: the use of classical R-genes supported by an effective backup system, use of known durable resistance, pyramiding of major genes to present multiple gene barrier, deployment of combinations of major gene resistance, use of partial/polygenic resistance, exploitation of tolerance and the use of biotechnological aids for durable resistance. According to Parlevliet[16], if non-durable major genes are used in combination it may be more difficult for the pathogen to build up races with a wider virulence spectrum (complex races). Such genes can be exploited in combination in two ways: 1) Through gene deployment 2) Through multiple gene barriers/gene pyramiding (physical combination in the same genotype).
Alternate approaches of gene deployment: Gene deployment is the guided distribution of genes in space and time[14]. Gould[17] divided the gene deployment strategies in to two broad categories as 1) spatial (gene deployment at the farm level, at the field level and at the region level) and 2) temporal.
Spatial gene deployment
Gene deployment at the field level: Spatial resistance gene deployment can also be done over a small geographic region such as within a single field. Several authors[18-21] have explained that if a cultivar consists of individual plants that differ from each other in their resistance, the resultant heterogeneous plant population should reduce the rate of epidemic development and prevent selection of a single compatible phenotype in the pathogen population. Gene deployment at the field level includes variety mixtures, species mixtures, multi lines and multi blends.
Variety mixtures: Variety mixture refers to a homogeneous, spatial mixture of different genotypes of one plant species in a field[22]. In some European countries it has become common to cultivate cultivar mixtures, i.e., equal mixture of seeds of 3 or 4 different cultivars with different powdery mildew resistance in barley[23]. According to Burdon[24] the concept of mixtures is to shield individuals carrying one particular resistance with a range of others carrying other resistance genes. Disease resistance in multilines and variety mixtures depends on slowing down the development of the best-adapted race on each component[23]. Mechanisms by which disease reductions are obtained in mixtures include a decrease in susceptible tissue and therefore a decrease in inoculum potential within the mixture, an increase in distance for spores to move from one susceptible plant to another, the physical barrier of resistant plants and possibly cross-protection whereby the defensive mechanisms of one component of the mixture may be activated by an a virulent isolate from another component[24]. Wolfe[23] reported that good mixtures of spring barley varieties resulted in up to 80% reduction in powdery mildew compared with mean disease levels of the components grown as pure stands. More ever cultivation of cultivar mixtures with different race specific resistance genes it is generally assumed that the effective lifetime of the resistance is prolonged and that partially defeated resistance genes may add significantly to the overall resistance of a mixture[25]. Even if variety mixtures prevent the evolution of virulent pathogens, mixtures will not be agriculturally useful if disease damage is high, as might happen if susceptible varieties are a large percentage of the mixture[22].
Variety mixtures have epidemiological advantage in that the interaction among neighboring plants in terms of spore dispersal leads to a reduction in disease spread relative to that in pure stands moreover cultivation of appropriate mixtures in an environment with a high disease risk provides a degree of high stability, which cannot be obtained from pure stands (buffering effects) another advantage is the use of cultivar mixtures ensures that the farmer does diversity among the cultivars available[13,23]. However, perhaps the biggest problem with mixtures is finding the correct combination of varieties, the fear as to the development of “super-races” and Therefore, mixtures are widely used in developing countries like Ethiopia where quality is not a priority.
Species mixtures/ mixed cropping: The safest opinion for mixed cropping, which may indeed provide permanent protection for cultivars with durable resistance, is to grow species mixtures in which the important pathogens are incompatible with one or more of the host species used. Of the many examples available, mixtures of barley and oat cultivars are widely used in cereal producing countries, for example in Canada and Poland, largely because of their great stability of production over years and regions[26]. Mixed cropping of cotton species is common in the Malwa plateau of central India[19]. Oat+barley mixtures have been suggested as an agro-technical method for controlling the oat sterile-dwarf virus disease. Finger millet+sorghum and Chickpea+safflower mixtures are common species mixture in Ethiopia.
Multiline varieties: The term multiline variety was first defined by Jensen[20] as a blend of multiple pure lines, each of which is of a different genotype. Borlaug[27] used an equivalent term ‘composite’ and proposed as a new approach for resistance to stem rust of wheat (Puccinia graminis tritici). According to Borlaug’s definition, these are mixtures of a number of phenotypically similar lines, which are genotypically different for resistance to a disease. These are mixtures of several pure lines of similar height, flowering and maturity dates, seed color and agronomic characteristics, each of which has a different gene for resistance to the given disease[3]. The term multiline in current usage designates a mixture of near-isogenic lines for resistance to a specific race of the pathogen[28].
Multilines have been known for mid-America at least since 1898[19]. Borlaug[18] suggested a minimum of 8 to 10 lines, thereby requiring 8 to 10 backcrossing programmes simultaneously and screening for genes resistant to a specific race that makes it damn expenses. Marshal[29] has given two approaches of multiline development. These are the clean crop approaches and the dirty crop approach. Dirty multilines are mixtures of lines each carrying a unique resistance gene and clean multilines are mixtures of lines each resistant to all prevalent races of the disease. Multiline varieties offer various advantages. For example, by using multilines we can exploit horizontal resistance, which is otherwise difficult to achieve because of the complexities associated with its genetic mechanism[27], multiline varieties proved to live longer than their corresponding pure stands because of stabilized selection. Another advantageous features of multilines over variety mixtures is that all the component lines are phenotypically similar the only difference being the gene for resistance.
Multiblend varieties/mass reservoirs: Multiblend varieties are generally simple mixtures of existing lines or varieties.
Gene deployment at the farm level (inter field diversification): A very large area of a single variety, or a number of varieties with similar resistance factors, would be vulnerable to any corresponding virulent isolate in the pathogen population[30]. A practical way to protect resistant factors, which the grower can understand and implement, is to diversify by growing a number of varieties, which differ in the form of their resistance to specific diseases[4]. However, a crucial point is that for diversification to be successful, we need more precise information on the differences among cultivars in terms of the genetics and mechanisms of their durability[26]. If the specific resistance genes in each variety selected differ, a pathogen race, which possesses specific virulence genes for one variety, will not spread to the other varieties[13]. Diversification is used primarily in cereal varieties for reduction of mildew and yellow rust.
Gene deployment at the regional level: If the cultivars recommended in different regions of an epidemiological area carry consistently different resistance genes it would certainly reduce the rate at which complex races would develop[14]. The probable success of regional gene deployment to suppress epidemics has good precedent[31]. Examples are found else where[19].
Temporal gene deployment: Temporal gene deployment may include 1) sequential release of resistance genes where by each variety is used until populations reach the breakdown population level and is immediately replaced by another variety[22]. The sequential release strategy has been employed for resistance to brown plant hopper (Nilaparvata lugens) at IRRI, 2) Variety rotations from season to season or recycling of resistance genes. Rotation of varieties with different resistances prevents selection of compatible isolates in populations of soil-born pathogens[31]. This approach was used to control stem rust of wheat in Australia between 1938 and 1950. According to Harahap and Silitonga[32] gene rotation would be effective only in areas of intensive agricultural production where plant pathologists maintain adequate and intensive disease surveys in cooperation with plant breeders.
Multiple gene barriers/ multigenes/ gene pyramids: An alternative to multilines is to pyramid desirable genes to provide a wide array of horizontal resistance[33]. In some areas although a single gene confers resistance to the existing pathogen population, the large-scale use of this gene results in the breakdown of resistance. To delay such breakdown, pyramiding of more than one resistance gene was found to be effective[15]. It is also known as gene stacking. Watson and Singh[34] proposed the concept of multigene varieties. Resistance gene pyramids are expected to considerably extend the durability of resistance because of the low probability for the pathogen to assemble multiple, rare virulence genes by mutation and/ or recombination[25]. In a way analogous to partial resistance breeding, the combining or pyramiding of resistance genes into individual cultivars has had considerable success in reducing the rate of evolution of pathogens particularly in situations where the pathogen does not reproduce sexually[24]. However some difficulties arise like for instance, in order to arrange this, one has to know all the resistance genes involved and the breeders in the whole epidemiological area should stick rigorously to the agreements made over the use of the resistance genes. Moreover, bringing together two fully effective major genes in an agronomically successful cultivar is far from easy[14]. However, in the future it is possible this may also be achieved by combinations of resistance genes that would require combinations of virulence genes that may have detrimental effects on pathogen fitness[24]. When sufficient RFLP markers have become available the application of a multiple gene barrier will become much easier as the tracing of the individual resistance gene becomes much easier[14].
Achievements of the various approaches of gene deployment: Two variety mixtures of spring barley, viz., Boss+Bryl+Rambo and Boss+Rabel+Rambo were recommended for production in Roslin for disease resistance and yield[35]. Several multiline varieties of various crops were released in different countries. Example, in USA., Ioa, E68, E69 and E70 of oat varieties[36], Miramar 63, Miramar 65 of wheat multiline varieties in Columbia for resistance to Stripe and Stem rusts[37], Peanut multilines, Florigiant (Sib-Lines), Florunner (Sib-Lines) in Virginia[38], Winter wheat multiline varieties resistant to yellow rust (P. striiformis) in the Netherlands[27]. In India wheat multiline varieties were developed that were resistant to Yellow rust and Brown rust. These were: Kalyansona lines KSML3, MLKS11, KML7406/Bithoor and Sonalika lines, MLSKA-9, MLSKA-12[3,28].
Several winter wheat cultivars remained resistant to stripe rust for periods of 15 or more years in Western Europe because of a multigene barrier/gene pyramiding[14]. DNA marker assisted selection was used to pyramid four Bacterial Blight (BB) resistance genes, Xa-4, xa-5, xa-13 and Xa-21 in rice[39]. According to these authors the pyramid rice lines showed a wider spectrum and a higher level of resistance than lines with only a single gene. Moreover, three Bacterial Blight (BB) resistance genes, xa5, xa13 and xa21 were successfully transferred to the New Plant Types (NPT) in rice[40] so that resistance would be long-lasting.
Plant breeders are always incorporating resistance genes into their otherwise high yielding but susceptible crop varieties. In so doing the major concern has been how to make the resistance genes last longer. Unless these genes are made durable, there will be always a need to make new incorporations which makes the operation uneconomical. A number of ways have been suggested by different authors to achieve durability of resistance of which gene deployment, the guided distribution of genes in space and time, is one. Although costly, the use of multilines is quite a better approach of gene deployment. Because of their poor uniformity variety mixtures can be used only in countries where quality is less important than yield. The recent approaches of gene deployment like pyramiding of different resistance genes into a common background that are carried out using molecular breeding guided back crossing have global prospects where there is access to carryout molecular analysis.
REFERENCES
- Houten, J.G.T., 1974. Plant pathology, changing agricultural methods and human society. Ann. Rev. Plant Pathol., 12: 1-11.
PubMedDirect Link - Tatum, L.A., 1971. The southern corn leaf blight epidemic. Science, 171: 1113-1116.
CrossRefDirect Link - Parry, D.W., 1990. Plant Pathology in Agriculture. Cambridge University Press, Cambridge, UK., ISBN-13: 978-0521363518, Pages: 402.
Direct Link - Gnanamanickam, S.S., V.B. Priyadarisini, N.N. Narayanan, P. Vasudevan and S. Kavitha, 1999. An overview of bacterial blight disease of rice and strategies for its management. Curr. Sci., 77: 1435-1444.
Direct Link - Browning, J.A. and K.J. Frey, 1969. Multiline cultivars as a means of disease control. Ann. Rev. Phytopathol., 7: 355-382.
CrossRefDirect Link - Wolfe, M.S., 1985. The current status and prospects of multiline cultivars and variety mixtures for disease resistance. Ann. Rev. Phytopathol., 23: 251-273.
Direct Link - Marshal, D.R., 1977. The advantages and hazards of genetic homogeneity. Ann. N. Y. Acad. Sci., 287: 1-20.
CrossRefDirect Link - Frey, K.J., J.A. Browning and M.D. Simons, 1977. Management systems for host genes to control disease loss. Ann. N. Y. Acad. Sci., 287: 255-274.
CrossRefDirect Link - Schilling, T.T., R.W. Mazingo, T.C. Wyne and T.G. Isleib, 1983. A comparison of peanut multilines and component lines across environments. Crop Sci., 27: 101-105.
Direct Link - Huang, N., E.R. Angeles, J. Domingo, G. Magpantay and S. Singh et al., 1997. Pyramiding of bacterial blight resistance genes in rice: Marker-assisted selection using RFLP and PCR. Theor. Applied Genet., 95: 313-320.
CrossRefDirect Link - Sanchez, A.C., D.S. Brar, N. Haung, Z. Li and G.S. Khush, 2000. Sequence tagged site marker-assisted selection for three bacterial blight resistance genes in rice. Crop Sci., 40: 792-797.
CrossRefDirect Link








