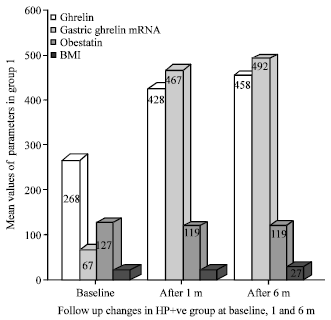
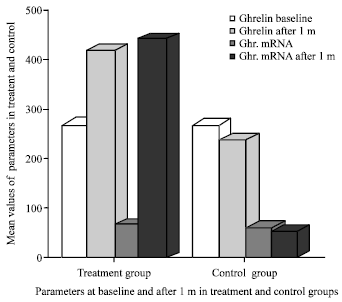
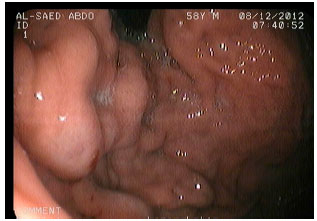
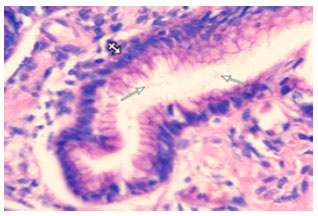
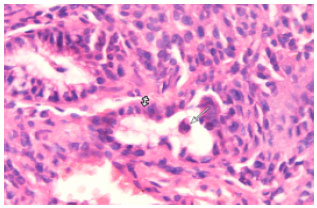
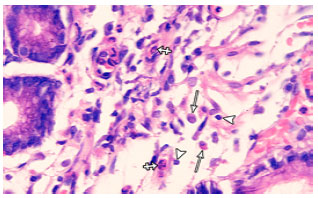
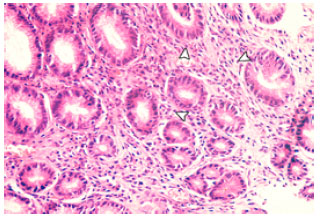
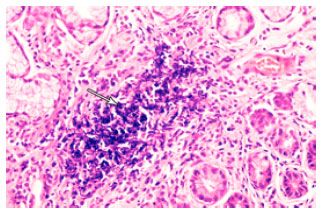
Research Article
Effect of H. pylori and its Eradication on Gastric Ghrelin Secretion
Department of Internal Medicine Gastroenterology and Hepatology Unit, Faculty of Medicine, Mansoura University, Egypt
Nesreen Moustafa Omar
Department of Histology and Cell Biology, Faculty of Medicine, Mansoura University, Egypt