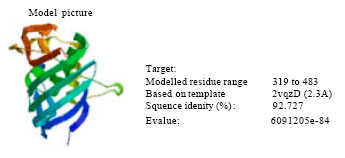
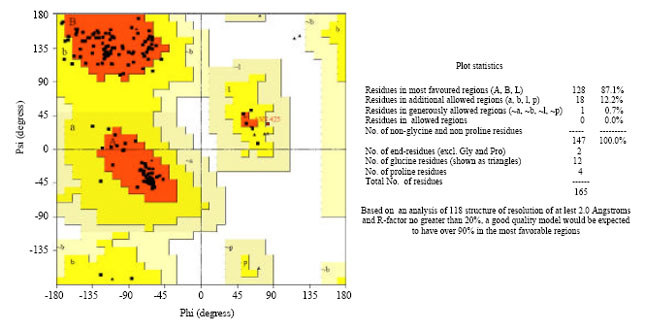
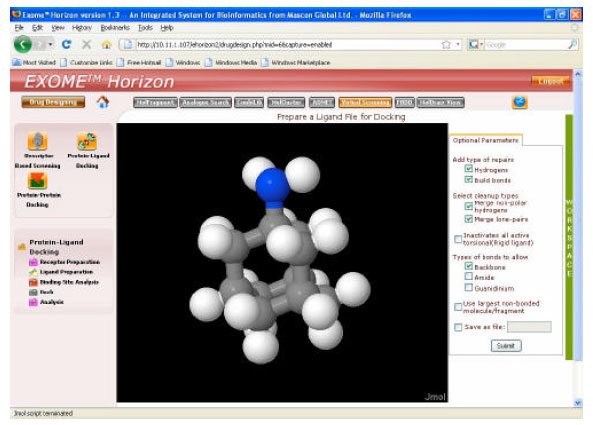
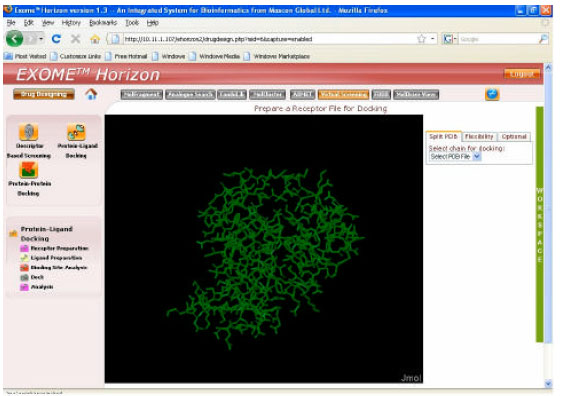
Research Article
In silico Molecular Docking of Influenza Virus (PB2) Protein to Check the Drug Efficacy
Department of Biotechnology, Meerut Institute of Engineering and Technology, N.H. 58, Delhi-Roorkee Highway, Baghpat Road Bypass Crossing,Meerut-250005, UP, India
M. A. Siddiqui
Department of Biotechnology, Meerut Institute of Engineering and Technology, N.H. 58, Delhi-Roorkee Highway, Baghpat Road Bypass Crossing,Meerut-250005, UP, India
N. Kapoor
Department of Biotechnology, Meerut Institute of Engineering and Technology, N.H. 58, Delhi-Roorkee Highway, Baghpat Road Bypass Crossing,Meerut-250005, UP, India
A. Arya
Department of Biotechnology, Meerut Institute of Engineering and Technology, N.H. 58, Delhi-Roorkee Highway, Baghpat Road Bypass Crossing,Meerut-250005, UP, India
H. Kumar
Department of Biotechnology, Meerut Institute of Engineering and Technology, N.H. 58, Delhi-Roorkee Highway, Baghpat Road Bypass Crossing,Meerut-250005, UP, India