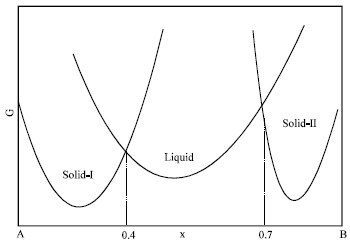
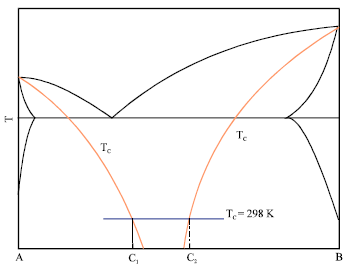
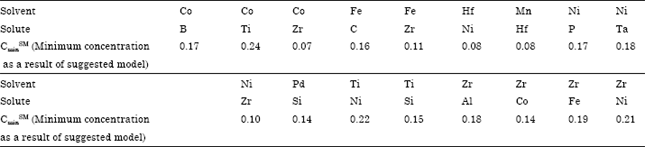
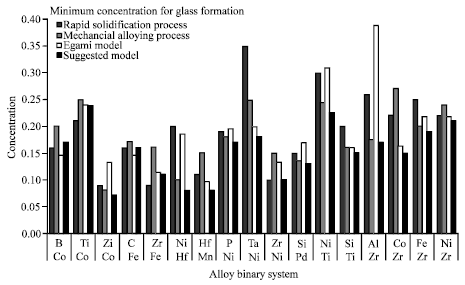
Research Article
A New Model in Glass Forming Range of Binary Alloys
Department of Materials Science and Engineering, School of Engineering, Shiraz University, Zand Blvd., 7134851154 Shiraz, Iran
M.J. Hadianfard
Department of Materials Science and Engineering, School of Engineering, Shiraz University, Zand Blvd., 7134851154 Shiraz, Iran