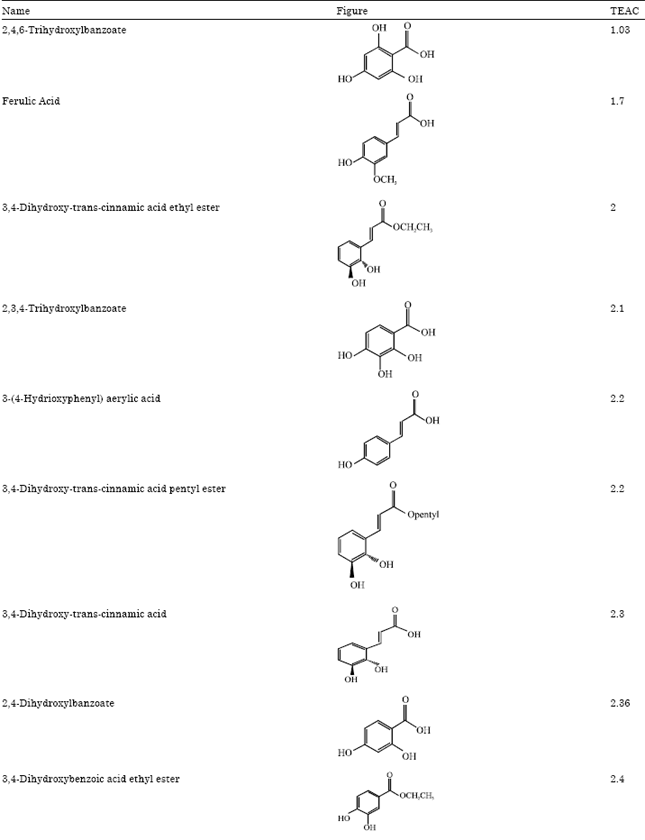
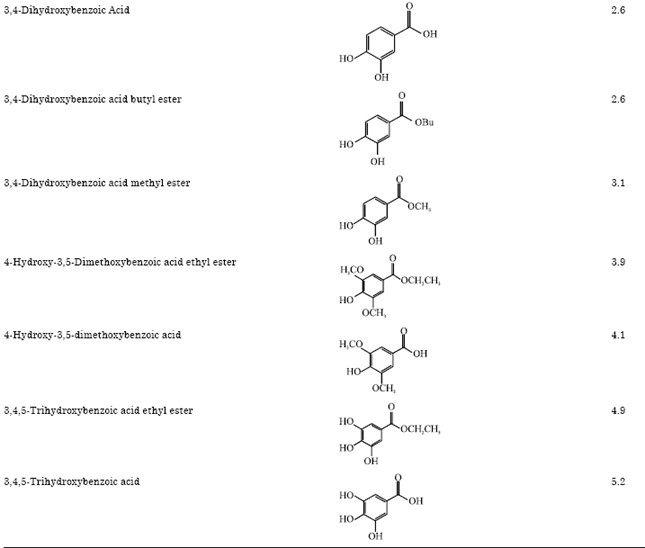
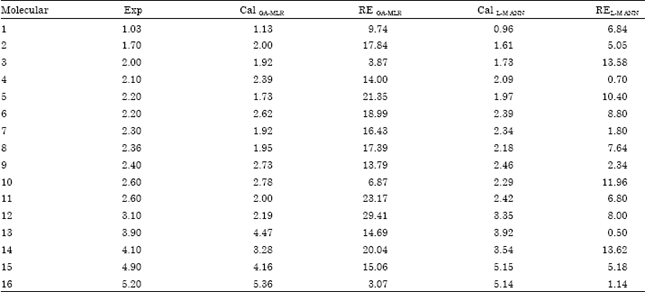
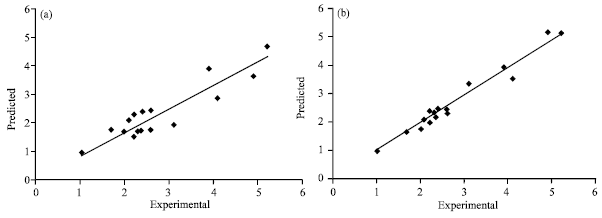
Research Article
Investigation of Capacity Behaviors by Linear and Nonlinear Models Chemometrics
Department of Chemistry, Faculty of Sciences, Arak Branch, Islamic Azad University, Arak, Iran
A. Farmany
Department of Chemistry, Faculty of Sciences, Arak Branch, Islamic Azad University, Arak, Iran