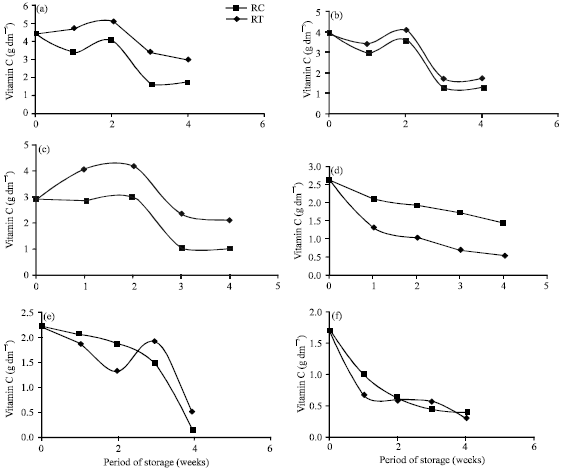
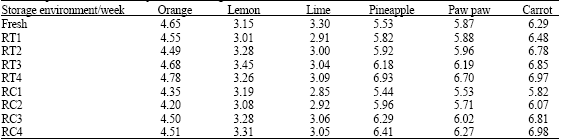
Research Article
The Effect of Storage Method on the Vitamin C Content in Some Tropical Fruit Juices
Department of Chemistry, Ahmadu Bello University, Zaria, Nigeria
O.A. Babatunde
Department of Chemistry, Nigerian Defence Academy, Kaduna, Nigeria
S. Suleiman
Department of Chemistry, Ahmadu Bello University, Zaria, Nigeria