Research Article
Proximate and Amino acid Analyses of Full-fat Sunflower (Helianthus annuus L.) Seed Meal
Animal Production Programme, School of Agriculture, Abubakar Tafawa Balewa, University, Bauchi, P.M.B. 0248, Bauchi State, Nigeria
Sunflower is an oilseed that is nutritious and unique in nature. A distinguishing feature of the sunflower is that, its flowering head tracks the sun’s movement. This occurrence is known as heliotropism. This property has been shown to increase light interception and possibly photosynthesis (Putnam et al., 1990).
The domesticated sunflower has a single-stem and a large seed head. This is in contrast to the wild variety that is highly branched with small heads and seeds. The sunflower head is made up of 1,000 to 2,000 individual flowers that are joined together to a base. The large petals that are found around the edge of a sunflower are individual ray flowers, these do not develop into seeds. Sunflower is not considered highly drought tolerant, but often produces satisfactory results when other crops are damaged during drought (Putnam et al., 1990).
Sunflower is one of the most important oil crops of major economic importance and ranks second to soybean among all oil seeds globally as a source of vegetable oil (Putt, 1978). Sunflower seed (full-fat) is grown mainly for its oil and to a lesser extent for the confectionery market (Olomu, 1995). Most United States production is devoted to the oil-seed type of sunflower, while a smaller percentage is grown for whole seed confectionery uses such as candy, snack food and baked foods. Sunflower seeds are commonly used as garnish or ingredients in many recipes, thickening soups, gravies and sauces and as part of salads and cereals. Sunflower seeds are also used to produce other things like medicine, paint, animal feed and bio-diesel (Putnam et al., 1990). Sunflower oil is considered premium oil due to it light colour, mild flavour, low level of saturated fats and ability to withstand high cooking temperatures (Putnam et al., 1990). Sunflower seed is rich in energy, since it has from 3,691 to 5,004 kcal of ME kg-1 and from 19.9 to 43.4% of ether extract (Daghir et al., 1980; Cheva-Isarakul and Tangtaweewipat, 1990; Tsuzuki et al., 2003). According to Olomu (1995) full-fat sunflower seed has crude protein of 17%, crude fat of 26.1%, crude fibre 29.1%, ash 3.1%, nitrogen free extract 18.8%, calcium 0.17% and total phophorus 0.53%. Tsuzuki et al. (2003) in their study reported that sunflower seeds contained; dry matter 93.10%, metabolizable energy 4,525 (kcal kg-1), crude protein 21.75%, ether extract 39.89%, crude fibre 15.51%, calcium 0.33% and total phosphorus 0.72%. Sunflower seeds are the best natural, whole food source of vitamin E, almost all of which is alpha-tocopherol, the most biologically active form (USDA Nutrient Database, 2002). Several authors have reported the use of sunflowers seeds in livestock diets. Sunflower seed meal has been incorporated in rations of poultry (Rose et al., 1972; Uwayjan et al., 1983; Kashani and Carlson, 1988; Karunajeewa et al., 1989; Jiang et al., 1991; Olomu, 1995; Villamide and Sanjuan, 1998; Tsuzuki et al., 2003; Adeniji and Ogunmodede, 2006), for rabbits (Akande et al., 2009) and swine (Olomu, 1995).
Nutritional data and information on sunflower seed are scanty and therefore this study was carried out to determine the proximate composition and amino acid profile of sunflower seeds as basis for evaluating its nutritional quality.
The experiment was carried out in November, 2006 in Bauchi, Bauchi town is located at latitude 13° 30’N and longitude 11° 50’E. Bauchi State is located in the Northern Guinea and Sudan Savanna zones of Nigeria.
Processing of sunflower seeds: The seeds of sunflower were roasted in a wide aluminium frying pan to the temperature of about 80°C. The seeds were mixed together by constant stirring to prevent the burning of the seed coat and enhance even distribution of heat. The roasting of sunflower seeds took 1-2 min. The seeds were allowed to cool and then milled in hammer mill.
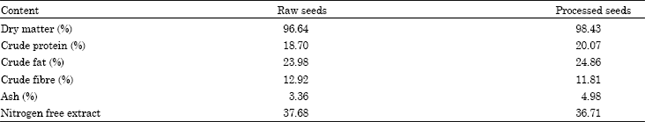
Proximate analysis: Proximate analyses of sunflower (both raw and processed seeds) were carried out using the methods outlined by the Association of Official Analytical Chemists (AOAC, 1990). The proximate compositions of the sunflower seeds are presented in Table 1.
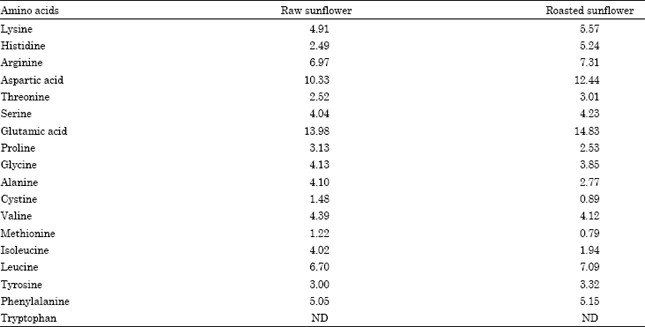
Amino acid analysis: The amino acid compositions of the samples were determined using the method described by Speckman et al. (1958). The samples were dried to constant weight and defatted. A known weight of the defatted sample was hydrolysed under vacuum with 7 mL of 6 N HCl in a sealed pyrex tube at 105°C for 22 h. Immediately after cooling, it was filtered through non-absorbent cotton wool. The filtrate was dried at 40°C using rotary evaporator.
The amino acids in the flask were diluted with 5 mL of acetate buffer (pH 2.0) and 5 to 10 μL was loaded into the cartridge of Technicon Sequential Multisample Amino acid Analyzer (TSM). The steam carrying the amino acid reagent mixture went through a heating bath where development of the coloured reaction product occurred. The absorbance was proportional to the concentration of each amino acid and was measured by colorimeter.
The proximate content of whole sunflower seed meal (both raw and roasted) is shown in Table 1. The range of values obtained from the proximate composition of sunflower seeds were; for dry matter (96.64-98.43%), crude protein (18.70-20.07%), crude fat (23.98-24.86%), crude fibre (11.81-12.92%), ash (3.36-4.98%) and nitrogen free extract (36.71-37.68%). The amino acid profiles of raw and roasted sunflower seeds are presented in Table 2. The results showed that the concentrations of most of the amino acids were actually increased with heat processing, with the exception of proline, glycine, alanine, cystine, valine, methionine and isoleucine. Glutamic acid had the highest concentration of 13.98 g/6 gN in the raw seeds and 14.83 g/16 gN in the roasted seeds. The most limiting amino acid was found to be methionine with values of 1.22 g/16 gN and 0.79 g/16 gN in the raw and roasted seeds, respectively. The second limiting amino acid was cystine (1.48 g/16 gN in the raw seed and 0.89 g/16 gN in the roasted seed). Arginine had the highest concentration among the indispensable amino acids with the value of 6.97 g/16 gN and 7.31 g/16 gN in the raw and roasted seeds, respectively.
| Table 1: | Proximate composition of raw and processed sunflower seeds |
 | |
| Table 2: | Amino acid profiles of raw and roasted sunflower seeds (g/16 g N) |
 | |
| ND: Not Determined | |
The concentrations of lysine in the sunflower seeds were also relatively high (4.91 g/16 gN for the raw seeds and 5.57 g/16 gN for the roasted seeds).
The chemical composition of sunflower seed depends on the weather, soil, variety and method of cultivation of the crop (Karunajeewa et al., 1989; Senkoylu and Dale, 1999). The range of values obtained from the proximate composition of sunflower seeds were; for dry matter (96.64-98.43%), crude protein (18.70-20.07%), crude fat (23.98-24.86%), crude fibre (11.81-12.92%), ash (3.36-4.98%) and nitrogen free extract (36.71-37.68%). The crude protein range of 18.70-20.07% obtained in this study is close to the mean value of 21.75% reported by Tsuzuki et al. (2003). The crude fat content of sunflower is high which indicates and confirms that it is a good source of vegetable oil. Olomu (1995) reported a crude fat of 26.1% for whole sunflower seeds which is slightly higher than the range reported for sunflower in this study. The difference in values reported for the crude fat may be due to type of processing method used before ether extraction.
The determination of amino acid composition is helpful in feed formulation, it is also an important parameter in the evaluation of protein quality. Amino acid compositions of sunflower seeds obtained in this study revealed that among the essential amino acids, arginine had the highest concentration with the values of 6.97 g/16 gN and 7.31 g/16 gN in the raw and roasted seeds, respectively. These values were closely followed by leucine with the value of 6.70 g/16 gN for the raw seeds and 7.09 g/16 gN for the roasted seeds. The concentrations of the sulphur-containing amino acids (methionine and cystine) were drastically decreased with heat processing. This is a reflection of the instability of these amino acids when subjected to heat treatment. The values of methionine were 1.22 g/16 gN and 0.79 g/16 gN and those of cystine were 1.48 g/16 gN and 0.89 g/16 gN in the raw and roasted seeds, respectively. These sulphur-containing amino acids were found to be the most limiting in sunflower seed. Similar reports were made by Olomu (1995), but it however, contradicts the findings of Vieira et al. (1992) who reported that lysine was the first limiting amino acid in broiler sunflower meal-based diets. According to Kashani and Carlson (1988) amino acid composition of sunflower seed is variable, with levels of lysine and methionine ranging from 0.56 to 0.66% and from 0.33 to 0.50%, respectively. The variation in the chemical composition of sunflower meal that has been reported and may be due to the method of processing and oil extraction (Karunajeewa et al., 1989; Vieira et al., 1992).
Future use of sunflower seed meal in livestock feeds is a possibility if properly exploited. Heat treatment of sunflower seeds should be carried out with caution so as to prevent burning of the seed coat. It is necessary to supplement sunflower seed meal based diets with the sulphur-containing amino acids (methionine and cystine) in order prevent nutritional deficiencies of these essential amino acids.