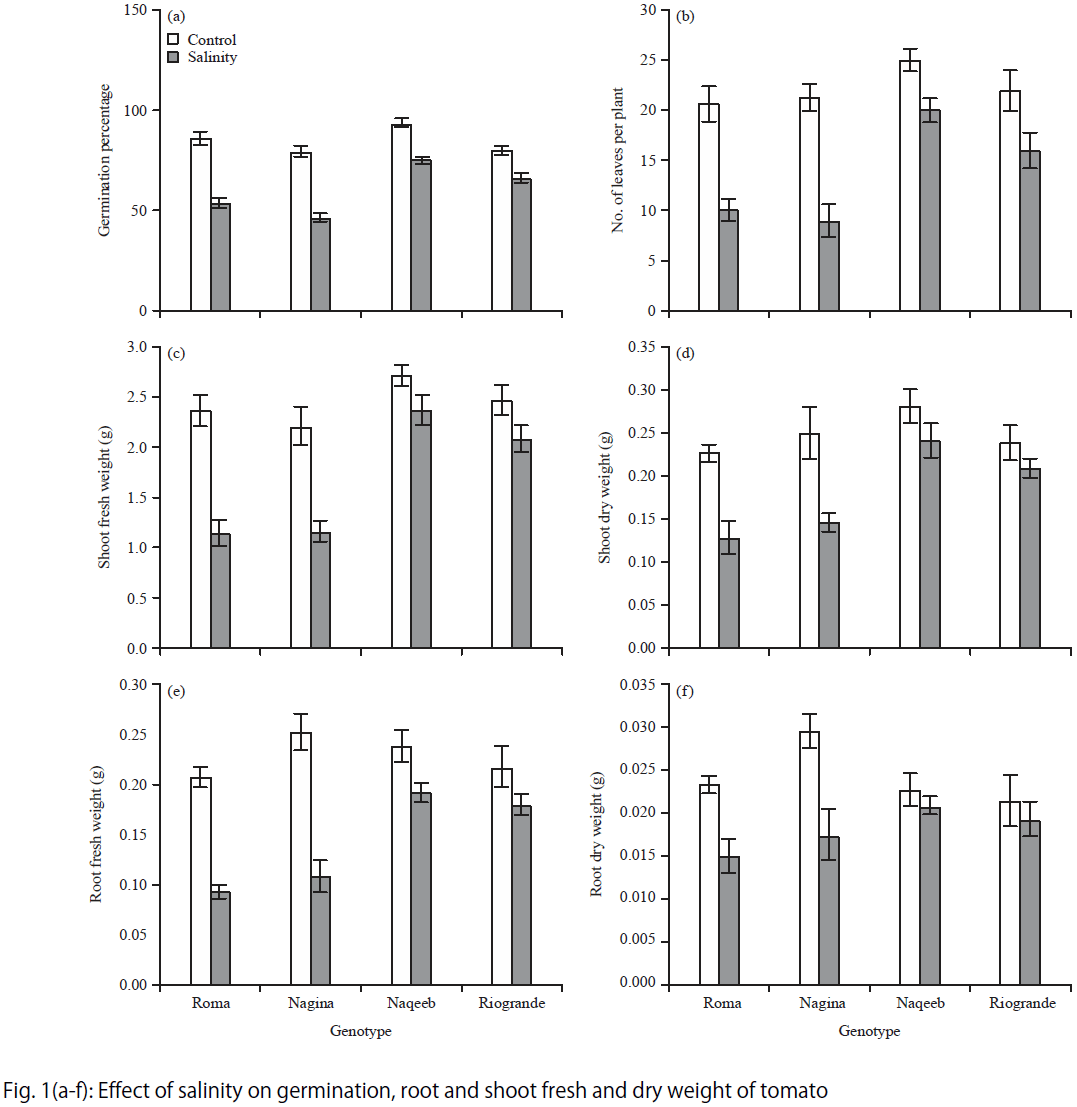
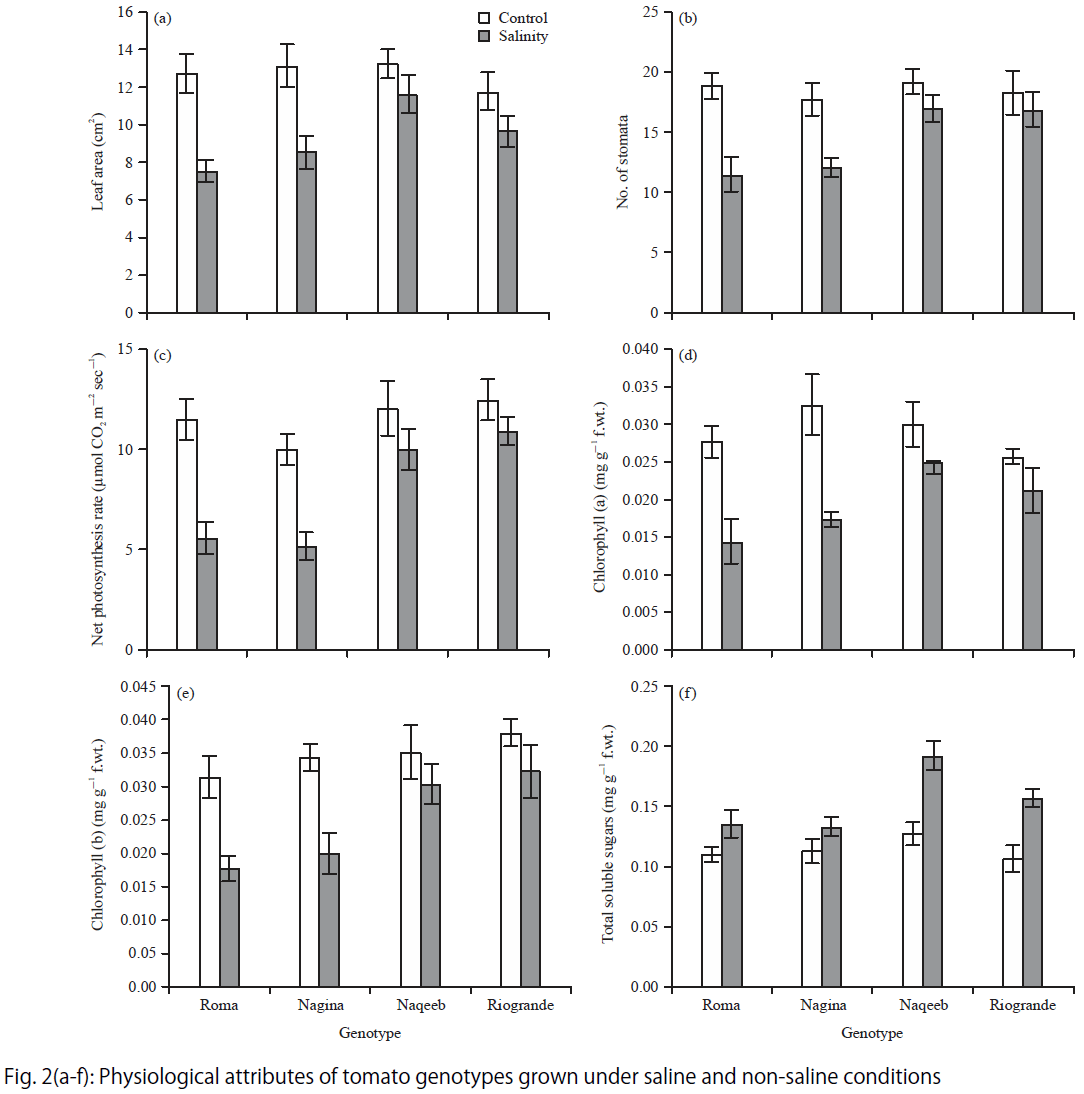
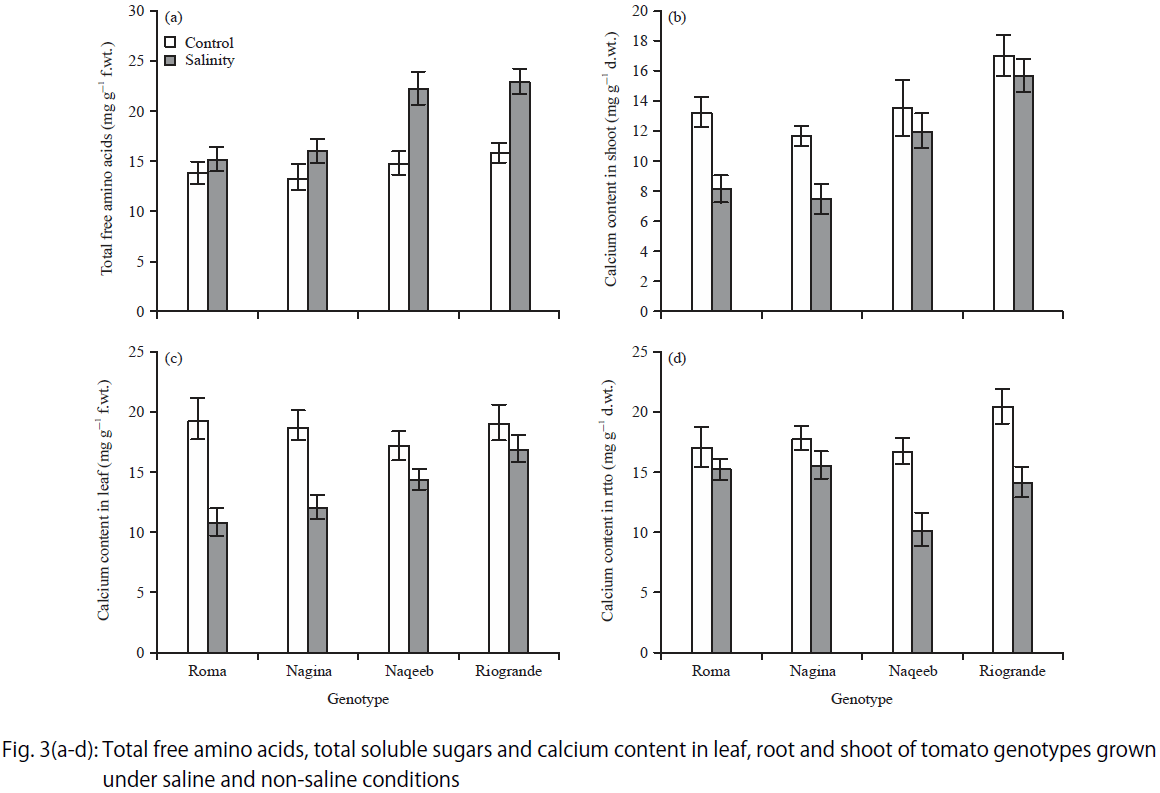
Research Article
Plant Growth is Highly Associated with the Concentration of Organic and Inorganic Osmolytes, Antioxidant Activities in Salt Stress Tomato (Lycopersicon esculentum) Plants
Department of Horticulture, University College of Agriculture, University of Sargodha, Sargodha-40100, Pakistan
Muhammad Mansoor Javaid
Department of Agronomy, University College of Agriculture, University of Sargodha, Sargodha-40100, Pakistan
Muhammad Adnan Shahid
Department of Horticulture, University College of Agriculture, University of Sargodha, Sargodha-40100, Pakistan
Hira Tariq
Nuclear Institute of Agriculture and Biology, Faisalabad, Pakistan
Muhammad Zubair
Department of Horticulture, University College of Agriculture, University of Sargodha, Sargodha-40100, Pakistan
Ahsan Akram
Department of Horticulture, University College of Agriculture, University of Sargodha, Sargodha-40100, Pakistan
Gulzar Akhtar
Department of Horticulture, University College of Agriculture, University of Sargodha, Sargodha-40100, Pakistan
Muhammad Wajid Khan
Department of Horticulture, University College of Agriculture, University of Sargodha, Sargodha-40100, Pakistan