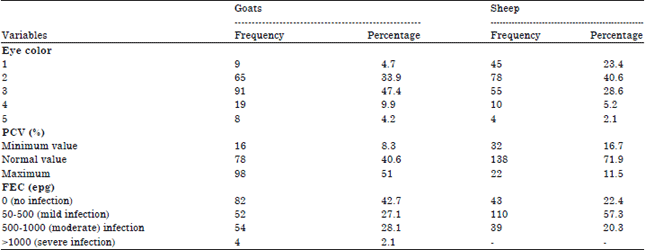
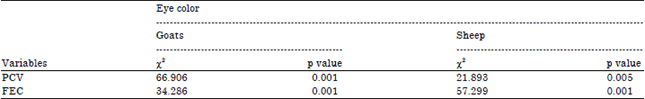
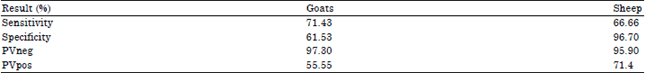
Research Article
Validation of the FAMACHA© Eye Color Technique for Detecting Anemic Sheep and Goats in Jigjiga Zone of Somali Region, Eastern Ethiopia
College of Veterinary Medicine, Jigjiga University, P.O. Box 1020, Jigjiga, Ethiopia
D. Dagnachew
College of Veterinary Medicine, Jigjiga University, P.O. Box 1020, Jigjiga, Ethiopia
Teshale Teklue
Mekelle Agricultural Research Center, P.O. Box 258, Mekelle, Ethiopia
N. Surendra
College of Veterinary Medicine, Jigjiga University, P.O. Box 1020, Jigjiga, Ethiopia