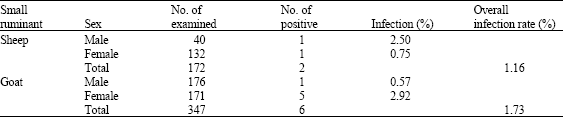Research Article
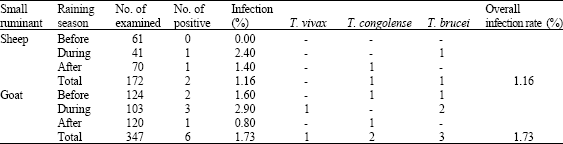
Periodic Variation in Trypanosoma Infection Rates in Trade Small Ruminants at Slaughter in Kaduna Central Abattoir
Nigerian Institute for Trypanosomiasis Research, P.M.B. 2077, Kaduna, Nigeria
A.O. Fajinmi
Nigerian Institute for Trypanosomiasis Research, P.M.B. 2077, Kaduna, Nigeria
J.O. Kalejaye
Nigerian Institute for Trypanosomiasis Research, P.M.B. 2077, Kaduna, Nigeria
B. Wayo
Nigerian Institute for Trypanosomiasis Research, P.M.B. 2077, Kaduna, Nigeria
M.K. Haruna
Nigerian Institute for Trypanosomiasis Research, P.M.B. 2077, Kaduna, Nigeria
J.E. Yarnap
Nigerian Institute for Trypanosomiasis Research, P.M.B. 2077, Kaduna, Nigeria
A.O. Usman
Nigerian Institute for Trypanosomiasis Research, P.M.B. 2077, Kaduna, Nigeria
S.M. Hamra
Nigerian Institute for Trypanosomiasis Research, P.M.B. 2077, Kaduna, Nigeria
A. Jijitar
Nigerian Institute for Trypanosomiasis Research, P.M.B. 2077, Kaduna, Nigeria
R. Ogunwale
Nigerian Institute for Trypanosomiasis Research, P.M.B. 2077, Kaduna, Nigeria
R. Bizi
Nigerian Institute for Trypanosomiasis Research, P.M.B. 2077, Kaduna, Nigeria
R.P. Ovbagbedia
Nigerian Institute for Trypanosomiasis Research, P.M.B. 2077, Kaduna, Nigeria
J.N. Abenga
Department of Veterinary Pathology and Microbiology, University of Agriculture, P.M.B. 2373, Makurdi, Nigeria