Research Article
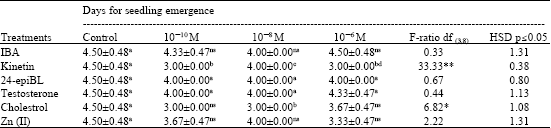
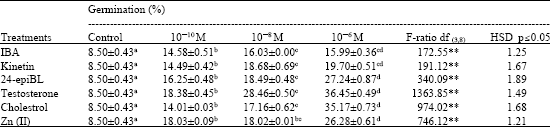
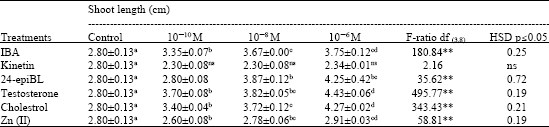
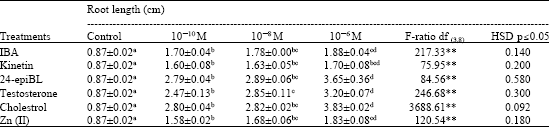
Enhancing Seed Germination of Chlorophytum borivilianum Sant. Et Fernand. with PGRs, Steroidal Hormones and Zinc
Department of Botanical and Environmental Sciences, Guru Nanak Dev University, Amritsar, 143005, Punjab, India
S. Arora
Department of Botanical and Environmental Sciences, Guru Nanak Dev University, Amritsar, 143005, Punjab, India
A.K. Thukral
Department of Botanical and Environmental Sciences, Guru Nanak Dev University, Amritsar, 143005, Punjab, India