Research Article
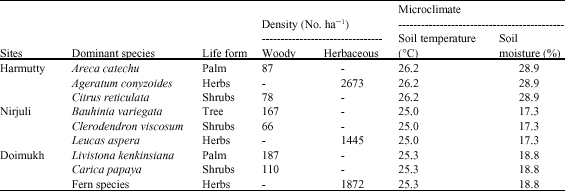
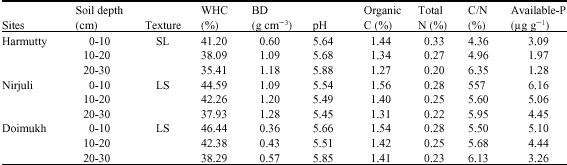
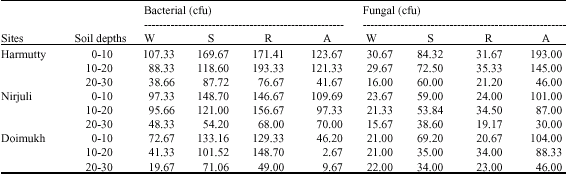
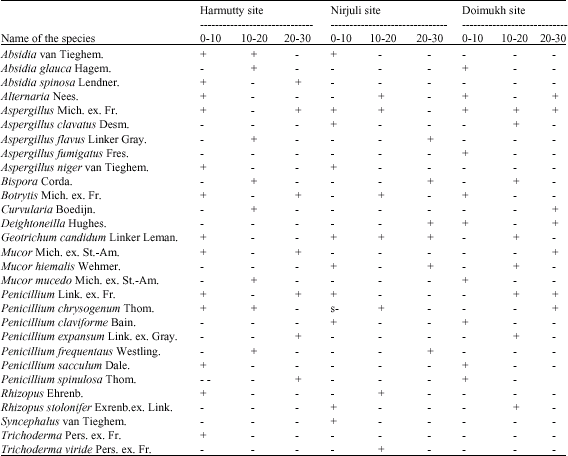
Microbial Population Dynamics of Soil Under Traditional Agroforestry Systems in Northeast India
Department of Botany, Rajiv Gandhi University, Rono Hills, Itanagar-791111, India
Kusum Arunachalam
Department of Forestry, NERIST, Nirjuli-791109, India
Ayyanadar Arunachalam
Department of Forestry, NERIST, Nirjuli-791109, India
Awadhesh Kumar Shukla
Department of Botany, Rajiv Gandhi University, Rono Hills, Itanagar-791111, India