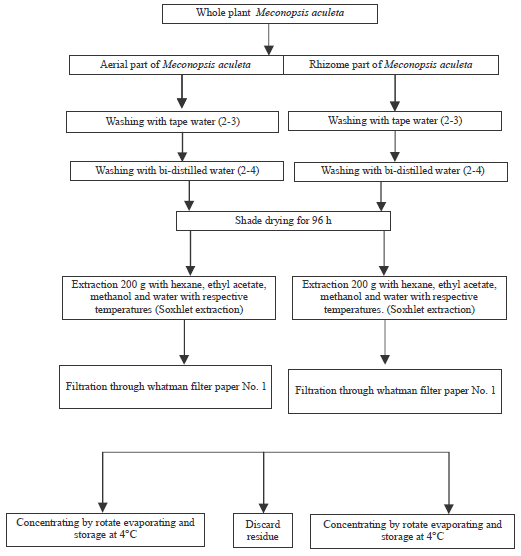
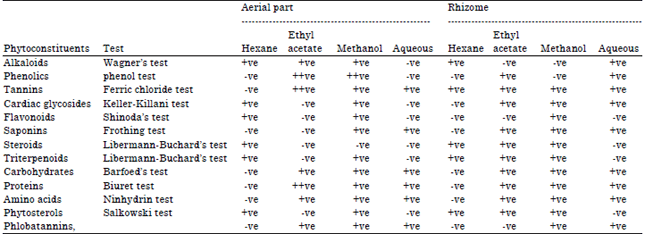
Perspective
Phytochemical Screening of Meconopsis aculeata Royle an Important Medicinal Plant of Kashmir Himalaya: A Perspective
Plant Tissue Culture Research Laboratory, Department of Botany, University of Kashmir, Srinagar-190 006, Jammu and Kashmir, India
LiveDNA: 91.14487
Zahoor A. Kaloo
Plant Tissue Culture Research Laboratory, Department of Botany, University of Kashmir, Srinagar-190 006, Jammu and Kashmir, India
Bashir A. Ganai
Phytochemistry Research Laboratory, Centre of Research for Development, University of Kashmir, Srinagar-190 006, Jammu and Kashmir, India
Hilal A. Ganaie
Phytochemistry Research Laboratory, Centre of Research for Development, University of Kashmir, Srinagar-190 006, Jammu and Kashmir, India
Seema Singh
Plant Tissue Culture Research Laboratory, Department of Botany, University of Kashmir, Srinagar-190 006, Jammu and Kashmir, India