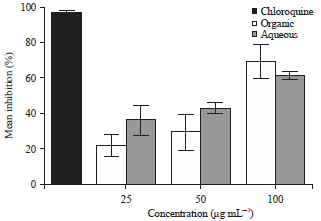
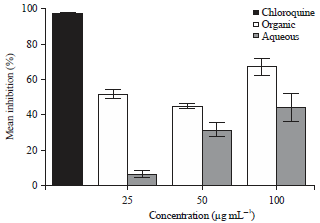
Research Article
Evaluation of the Antiplasmodial Properties of Namibian Medicinal Plant Species, Moringa ovalifolia
Multidisciplinary Research Centre, University of Namibia, Windhoek, Namibia
LiveDNA: 264.16196
Rahl-Jeanne Bussel
School of Pharmacy, University of Namibia, Windhoek, Namibia
Davis Mumbengegwi
Multidisciplinary Research Centre, University of Namibia, Windhoek, Namibia