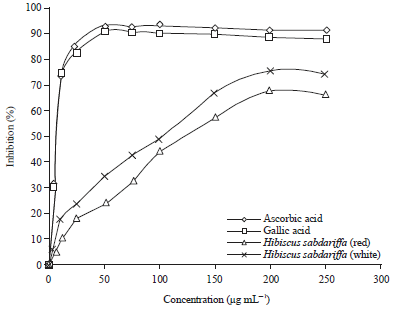
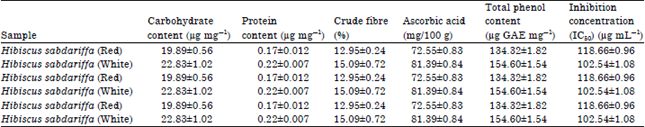
Research Article
Nutritional Evaluation of Calyces and Epi-Calyces from Two Variants of Hibiscus sabdariffa L.: A Comparison
Department of Biotechnology, Institutional Biotech Hub, Pandu College, Pandu, Guwahati, Assam, India
Pranjal Sarmah
Department of Biotechnology, Institutional Biotech Hub, Pandu College, Pandu, Guwahati, Assam, India
Aniruddha Sarma
Department of Biotechnology, Institutional Biotech Hub, Pandu College, Pandu, Guwahati, Assam, India