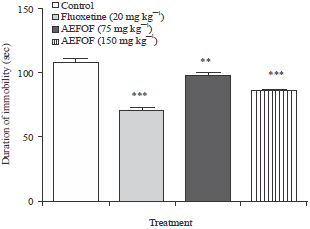
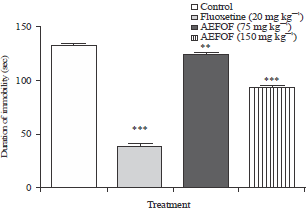
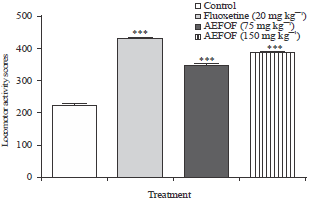
Research Article
Antidepressant Activity of Alcoholic Extract of the Fruits of Osmanthus fragrans Lour in Mice
Department of Pharmacology, Hygia Institute of Pharmaceutical Education and Research, Ghaila Road, Lucknow, 226020, Uttar Pradesh, India
Roli Gupta
Department of Pharmacology, Hygia Institute of Pharmaceutical Education and Research, Ghaila Road, Lucknow, 226020, Uttar Pradesh, India
Md. Azizur Rahman
Faculty of Pharmacy, Integral University, Dasauli, Kursi Road, Lucknow, 226026, Uttar Pradesh, India
Mehnaz Kamal
Faculty of Pharmacy, Integral University, Dasauli, Kursi Road, Lucknow, 226026, Uttar Pradesh, India