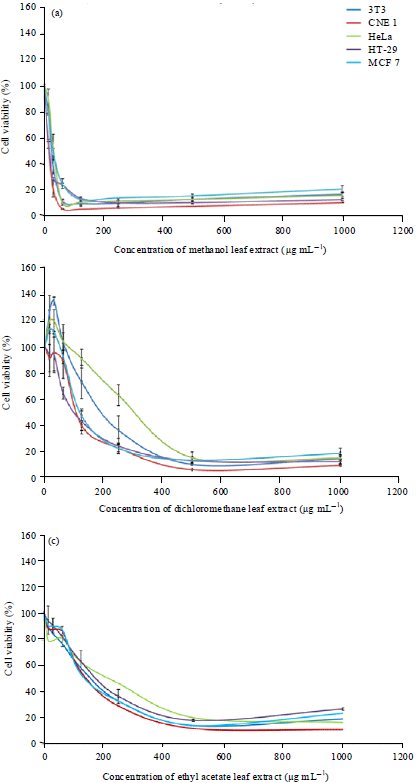
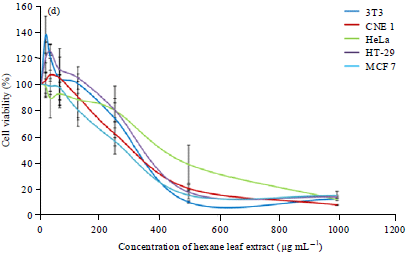
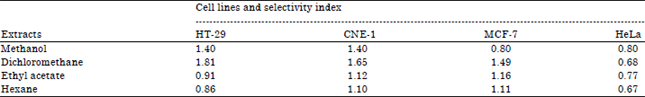
Research Article
Anti-Proliferative Activity and Preliminary Phytochemical Screening of Ipomoea quamoclit Leaf Extracts
International Medical University, No. 126, Jalan Jalil Perkasa 19, Bukit Jalil, Kuala Lumpur, 57000, Malaysia
Wei Ern Chung
International Medical University, No. 126, Jalan Jalil Perkasa 19, Bukit Jalil, Kuala Lumpur, 57000, Malaysia
Kah Ee Choong
International Medical University, No. 126, Jalan Jalil Perkasa 19, Bukit Jalil, Kuala Lumpur, 57000, Malaysia
Yan Li Cheah
International Medical University, No. 126, Jalan Jalil Perkasa 19, Bukit Jalil, Kuala Lumpur, 57000, Malaysia
Ee Ying Phua
International Medical University, No. 126, Jalan Jalil Perkasa 19, Bukit Jalil, Kuala Lumpur, 57000, Malaysia
Ramamurthy Srinivasan
International Medical University, No. 126, Jalan Jalil Perkasa 19, Bukit Jalil, Kuala Lumpur, 57000, Malaysia