Research Article
Evaluation for Antioxidant Activity of Artemisia sp. Plants
Department of Food and Nutrition, Kongju National University, Yesan, Chungnam, 340-702, Korea
Artemisia species, belonging to Compositae family, are distributed widely in Northern Hemisphere and approximately 25 species of Artemisia are distributed in Korea (Lee, 2006). Most species of Artemisia have strong aroma and bitter taste due to terpenoids and sesquiterpene lactones (Pace et al., 2010).
Several terpenoids, flavonoids, coumarins, acetylenes, caffeoylquinic acids and sterols are isolated from Artemisia species and are reported to have anti-malarial, antiviral, anti-tumor, anti-pyretic, anti-hemorrhagic, anti-coagulant, anti-anginal, antioxidant, anti-hepatitis, anti-ulcerogenic, antispasmodic and anti-complementary activities (Tan et al., 1998). Biological activities of Artemisia scoparia, Artemisia japonica, Artemisia capillaries and Artemisia montana have been studied diversely among Artemisia species. However, little is known about the bioactivity of Artemisia stolonifera, Artemisia keiskeana, Artemisia selengensis and Artemisia sylvatica. Artemisia sp. plants have medicinal importance and have been used as a traditional folk medicine for the treatment of a variety of diseases (Rho and Seo, 1993).
Reactive Oxygen species (ROS) are the chemically reactive ions and are generated as byproducts of primary metabolic activities in a body. Excess of ROS/free radicals damages the cellular enzymes, oxidizes the carbohydrates, proteins, lipids and DNA and causes the diseases and cellular injuries (Halliwell and Gutteridge, 1999). The ROS-mediated cellular injury has aroused the attention on natural antioxidants that can be supplemented as the dietary foodstuffs (Singh et al., 2008).
Although, several Artemisia sp. plants have been used as folk medicine for a long time in Korea, little is known about scientific evidences to explain their physiological activities. In this study, the antioxidant activity of eight Artemisia sp. plants is investigated for providing the evidences for theirs physiological antioxidant actions.
Chemicals and reagents: Diphenyl-1-picryl hydrazyl (DPPH), ascorbic acid, butylated hydroxytoluene (BHT), pyrogallol, 2,2'-azinobis-(3-ethyl benzothiazoline-6-sulphonic acid) (ABTS), 2,2'-azobis (2-amidio-propane) dihydrochloride (AAPH), 6-hydroxy-2,5,7,8-etramethylchroman-2-carboxylic acid (Trolox), fluorescein disodium salt, H2O2, CoF2•4H2O and picolinic acid were purchased from Sigma-Aldrich (St. Louis, MO, USA). Ethanol and other reagents were used as first grade reagents.
Plant materials: Extract of Artemisia sp. plants was obtained from Plant Extract Bank (PEB), Korea Research Institute of Bioscience and Biotechnology (KRIBB) (Daejeon, Korea). Artemisia sp. plants were authenticated and deposited by PEB, KRIBB; Artemisia japonica (Collection No. 009-077), Artemisia capillaries (Collection No. 016-057), Artemisia montana (Collection No. 012-055), Artemisia scoparia (Collection No. 034-042), Artemisia stolonifera (Collection No. 007-021), Artemisia keiskeana (Collection No. 032-024), Artemisia selengensis (Collection No. 008-040) and Artemisia sylvatica (Collection No. 017-021). Dried and crushed Artemisia sp. plants were extracted with methanol for 48 h at room temperature. Extracts were filtered through the defatted cotton and were concentrated by evaporator under reduced pressure. Artemisia sp. plant extracts were resolved in DMSO to 100 mg mL¯1, stored at -20° and used it as a stock.
Determination of total phenolic content and total flavonoid content: The total phenolic content was determined according to the Folin-Ciocalteu method with some modification (Oh et al., 2004), using gallic acid as a standard. Briefly, the extract was dissolved in 1 mL of 1 N Folin-Ciocalteu reagent. After adding 2 mL of 20% Na2CO3, the mixture was allowed to stand for 10 min. The mixture was centrifuged for 8 min and the absorbance of the supernatant was measured on 765 nm by a spectrophotometer (SpectraMax M2, Molecular Devices Inc., USA). Total phenolic content of extracts was expressed as mg gallic acid equivalents (GAE)/g dry weight (DW).
Total flavonoid content was measured by a modified Zhishen et al. (1999) protocol. Distilled water (1.25 mL) containing extract were mixed with 75 μL of 5% NaNO2 and was allowed to stand for 5 min. Then, 0.15 mL of 10% AlCl3•6H2O was added. After 6 min incubation in room temperature, 0.5 mL of 1 M NaOH and 0.275 mL distilled water were added. The absorbance of the mixture was measured on 510 nm using a UV-spectrophotometer (SpectraMax M2, Molecular Devices Inc., USA) and compared to a standard curve of catechin. Total flavonoid content of extract was expressed as mg catechin equivalents (CE)/g dry weight.
DPPH radical scavenging activity: Scavenging effect of Artemisia sp. plant extracts on 2,2-diphenyl-1-picrylhydrazyl (DPPH) free radical was measured by a following procedure. Briefly, the methanol (4.5 mL) containing extract was mixed with 0.5 mL of DPPH solution (1 mmol L¯1). The mixture was mixed for 5 sec, left to stand at room temperature for 30 min and the absorbance was read on 517 nm using UV-spectrophotometry (Agilent Technologies Inc., CA, USA).
Measurement of reducing power: The reducing power of Artemisia sp. plant extracts was determined by Fe3+ reduction. Artemisia sp. plant extracts were mixed with 2.5 mL of 0.2 M phosphate buffer (pH 6.6) and 2.5 mL of 1% K3Fe(CN)6. The mixture was incubated at 50°C for 20 min. Then 2.5 mL of 10% trichloroacetic acid was added and centrifuged at 2,090×g for 10 min. A 2.5 mL of supernatant layer was added to 2.5 mL of distilled water and 0.5 mL of 0.1% FeCl3. The absorbance of the mixture was measured on 700 nm using UV-spectrophotometry (Agilent Technologies Inc., CA, USA).
Nitric oxide (NO) radical scavenging assay: Nirtic oxide (NO) radical scavenging activity of Artemisia sp. plant extracts was measured by Babu et al. (2001) protocol with a little modification. Sodium nitroprusside, a chemical NO donor, generates NO radical, when interacted with oxygen in aqueous solution at physiological pH condition. Content of NO radical was measured by Griess reaction. Briefly, a reaction mixture (3 mL) containing sodium nitroprusside (10 mM in PBS) and samples were incubated at 25°C for 150 min. After incubation, 0.5 mL of the reaction mixture and 0.5 mL of Griess reagent (1% sulfanilamide in 5% phosphoric acid and 0.1% N-1-naphthylethylenediamine dihydrochloride in distilled water) were mixed. Intensity of the chromophore formed was detected on 540 nm.
ABTS radical cation assay: Scavenging of ABTS+• free radical by Artemisia sp. plant extracts was monitored by modified methods of Thaipong et al. (2006) and Gramza et al. (2005). To generate ABTS+• in phosphate-buffered saline (pH 7.4), the stock solution was prepared to have 7 mM ABTS and 2.45 mM potassium persulfate and was allowed to react for 24 h at room temperature in the dark. Then the dark blue-green colored ABTS+• radical solution was diluted to obtain an absorbance of 0.70±0.02 on 732 nm using the spectrophotometer (SpectraMax M2, Molecular Devices Inc., USA). Fresh ABTS+• solution was prepared for an each assay. Artemisia sp. plant-extract (10 μL) was reacted with 190 μL of the ABTS+• solution for 30 min in a dark condition. Absorbance of the reactant was determined on 734 nm using the spectrophotometer. Trolox was used as a standard for ABTS assay (0-1 mmol L¯1). Results were expressed as μmol Trolox Equivalents (TE)/g dry weight by comparison to the slope for ABTS+• scavenging by Trolox.
Oxygen Radical Absorbance Capacity (ORAC) assay: The ORAC assay was performed using the modified methods of Ciz et al. (2010) and Huang et al. (2002). The ORAC assay monitored the antioxidant scavenging function against peroxyl radical induced by AAPH at 37°C. Fluorescein was used as a fluorescent probe for antioxidant activity. The loss of fluorescence for fluorescein indicated the inhibition of the peroxyl radical production (Gomes et al., 2005). Fluorescein (70 nM) and other reagents were prepared in phosphate buffer (75 mM, pH 7.4). A 170 μL of fluorescein solution (60 nM final concentration) and 10 μL of sample were placed in a well of microplate (clear bottom, black plate) and were incubated at 37°C directly for 30 min. After the incubation, 20 μL of AAPH (50 mM, final concentration) was added rapidly using a multichannel pipette to start the reaction. The fluorescence was recorded every 5 min and the microplate was shaken automatically prior to each reading. Phosphate buffer was used as a blank and Trolox (3.125, 6.25, 12.5 and 25 μg mL¯1) was used as an antioxidant standard for plotting the calibration curve to quantitate oxygen radical absorbance capacity in each assay. Final ORAC values were calculated using a regression equation between the Trolox concentration and the net Area Under the Curve (AUC). Net AUC corresponding to the sample was calculated by subtracting the AUC for the blank. The ORAC values were expressed as μmol Trolox Equivalents (TE) per gram of Dry Weight (DW) of Artemisia sp. plant-extract.
Hydroxyl Radical Antioxidant Capacity (HORAC) assay: The HORAC assay measures the metal-chelating activity of antioxidants in the conditions of Fenton-like reactions employing a Co (II) complex and hence the protecting ability against formation of hydroxyl radical (Ou et al., 2002). A 4.6 mM Co(II) was prepared by dissolving 15.7 mg of CoF2•4H2O and 20 mg of picolinic acid in 20 mL of distilled water. A 170 μL of fluorescein (60 nM, final concentration) and 10 μL of sample were incubated in ELISA plate reader (SpectraMax M2, Molecular Devices Inc., USA) at 37°C for 10 min. After incubation, a 10 μL of 0.55 M H2O2 (27.5 mM, final concentration) and 10 μL of Co (II) (230 μM, final concentration) were added subsequently. The initial fluorescence was measured every minute after shaking. Phosphate buffer solution was used for a blank. Gallic acid solutions (100, 200, 600, 800 and 1000 μM) in 75 mM phosphate buffer (pH 7.4) were used for plotting the standard curve. The AUC were calculated by the same method to ORAC assay. Final HORAC values were calculated using a regression equation between the gallic acid concenstration and the net area under the curve. One HORAC unit was assigned to the net protection area, provided by 1 μM gallic acid and the activity of the sample was expressed as μmol Gallic Acid Equivalents (GAE) per gram of fresh weight of the samples.
Statistical assay: All results were expressed as a Mean±Standard Deviation (SD) of triplicate and were analyzed using one way Analysis of Variance (ANOVA) and Dunnett’s multiple comparison test for individual comparisons. Results were considered significant statistically, when p-values were p<0.05.
Total phenolic compounds and flavonoids contents of Artemisia sp. plants-extract: Content of total phenolic compounds and total flavonoids in eight Artemisia sp. plant extracts were shown in Fig. 1. Artemisia capillaries-extract had the highest value of the total phenolic content (120 mg GAE/g DW). Other Artemisia sp. plant extracts had approximately 66-101 mg GAE/g DW of the total phenolic content which means the phenolic compounds exist in approximately 6-12% of extract-solid material.
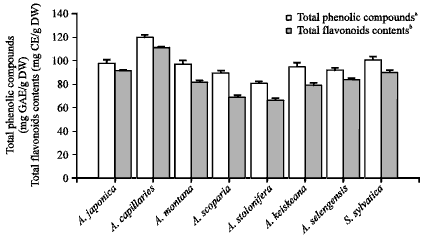 | |
| Fig. 1: | Total phenolic and total flavonoids contents of Artemisia sp. plants extract. aTotal phenolic contents are expressed as gallic acid equivalents (GAE; milligrams of gallic acid per gram of dried extract). bTotal flavonoid contents are expressed as catechin equivalents (CE; milligrams of catechin per gram of dried extract). Error bars have been omitted when the standard deviation was less than the size of the symbol |
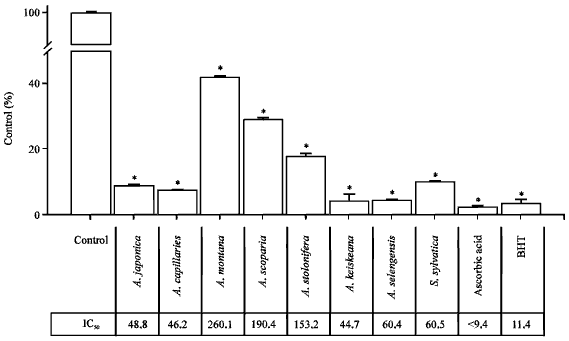 | |
| Fig. 2: | Scavenging effect of Artemisia sp. plants-extract on DPPH radicals. A 0.2 mL of methanolic solution containing extract (300 μg mL¯1) was mixed with 4 mL of methanol and DPPH (1 mmol L¯1, 0.5 mL) was added and left to stand at room temperature for 30 min and the absorbance read at 517 nm |
In addition, A. capillaries extract had the highest value of total flavonoids (approximately 111 mg CE/g DW), that means approximately 92.5% of flavonoids compounds in total phenolic contents. All Artemisia sp. plant extracts used in this study had approximately 77-93% flavonoids compounds in total phenolic contents.
Antioxidant activity of plant extracts is caused mainly from their phenolic compounds/flavonoids. Phenolic compounds have diverse structures and molecular weight in plants. The phenolic hydroxyl group of phenolic compounds can bind easily to macromolecules, such as proteins, in the extract. The amount of total phenolic compounds/flavonoid compounds play an important role for antioxidant, anticancer, anti-inflammatory and antimicrobial activity (Kosmider and Osiecka, 2004).
Antioxidant activity of Artemisia sp. plants-extract
DPPH radical scavenging activity: Authentic antioxidant ascorbic acid (9.4 μg mL¯1) and BHT (75 μg mL¯1) showed approximately 97.7±2.43 and 96.5±0.7% of DPPH radical scavenging activity (Fig. 2). Also the synthetic antioxidant BHT had a very low IC50 value (11.4 μg mL¯1) for DPPH radical scavenging but has been prohibited to use in foods, due to its bad side-effect. Therefore, many researchers are trying to explore the natural antioxidant materials which are safe to human body and available to use in food production. Eight Artemisia sp. plants in this study had the potent hydrogen-donating activity to DPPH radical in a concentration-dependent manner. The highest DPPH radical scavenging activity was found in A. keiskeana and A. selengensis with 95.7% (IC50 = 19.67) and 95.4% (IC50 = 28.7 μg mL¯1) and also A. capillaries and A. japonica had potent DPPH radical scavenging activities (92.2 and 91.2%), respectively. Artemisia japonica, A. capillairies and A. sylvatica showed relative high DPPH radical quenching activity, whereas A. montana, A. scoparia and A. stolonifera had low DPPH radical scavenging activity. Generally, antioxidant pattern of extract is reflected with its total phenolic/flavonoids contents. However, A. keiskeana and A. selengensis did not have the highest total phenolic/flavonoids contents among tested Artemisia sp. plants, these plants-extract showed the highest DPPH radical scavenging activity. Additionally, A. montana and A. scoparia had relatively low DPPH radical scavenging activity, compared with their content of total phenolic compounds.
Reducing power capacity: Additionally the reducing power of Artemisia sp. plants-extract increased in a concentration-dependent manner (Fig. 3). Pyrogallol, a positive control, had the potent reducing power A700 = 0.348±0.003 at 18.75 μg mL¯1. Artemisia sp. plant extracts had the reducing power A700 = 0.116-0.280, 300 μg mL¯1 which had the linearity and the consistency to DPPH radical scavenging activity totally. The slopes of reducing powers showed the highest value (7.44E-4) in A. japonica-extract and the lowest value (1.94E-4) in A. montana-extract. Artemisia japonica extract had 3.83 times more reducing capacity than that of A. montana-extract. It is anticipated that the free radical scavenging activity and reducing power of Artemisia sp. plants-extract exert the donating electron and can react with free radicals to convert them to more stable products and terminate radical chain reactions and help the relief on inflammatory symptoms, caused by harmful radical compounds.
In inflammation, the cell and tissue injury is due to the toxicity of ROS generated and released from activated phagocytes (Leirisalo-Repo et al., 1993). The NADPH oxidase of phagocytes is involved in production of superoxide anion and the proper function of phagocytes play an important role for primary immunity mechanism against invading microorganisms. Free radical scavenging activity and reducing power of Artemisia sp. plants-extract may play the critical role for quenching superoxide anions and maintaining the cellular redox homeostasis against harmful oxidants and free radicals in cells. Because the reducing power has been shown to exert an antioxidant effect by donating a hydrogen atom and breaking the free radical chain (Duh, 1998), it is associated closely with the presence of reductones which are strong reducing agents, thus efficacious antioxidants and fairly strong acids.
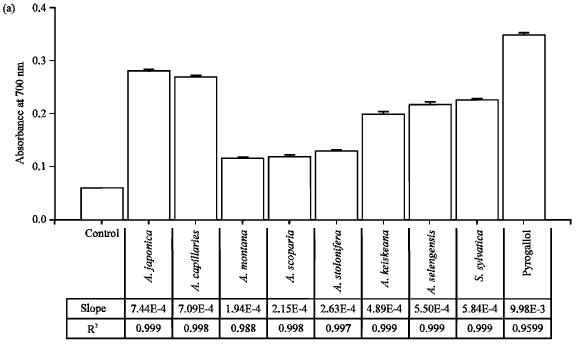 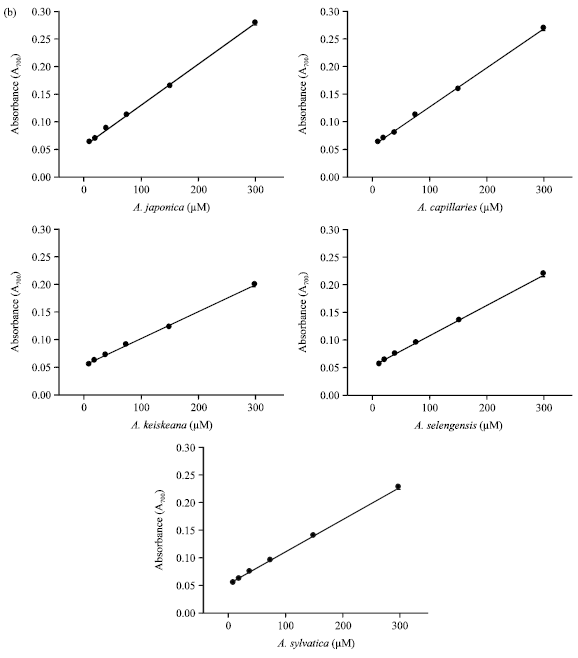 | |
| Fig. 3(a-b): | Reducing power of Artemisia sp. plants-extract. Artemisia sp. plants-extract (300 μg mL¯1) was mixed with 2.5 mL of 0.2 M phosphate buffer (pH 6.6) and 2.5 mL of 1% K3Fe(CN)6. The mixture was incubated at 50°C for 20 min. After that, 2.5 mL of 10% trichloroacetic acid was added and centrifuged. A 2.5 mL of supernatant layer was added to 2.5 mL of distilled water and 0.5 mL of 0.1% FeCl3. The absorbance was measured at 700 nm. Error bars have been omitted when the standard deviation was less than the size of the symbol, (a) Reducing power of Artemisia sp. plants (300 μg mL¯1). Slope and R2 are derived from plots (reducing power vs. concentration) and (b) Plots of reducing power for A. japonica, A. capillaries, A. kerskeana, A. selengensis and A. sylvatica |
NO radical scavenging activity: Scavenging activity of Artemisia sp. plant extracts for nitric oxide (NO) radical was investigated using sodium nitroprusside which is a chemical NO donor, produces nitrite ions in aqueous solution at physiological pH.
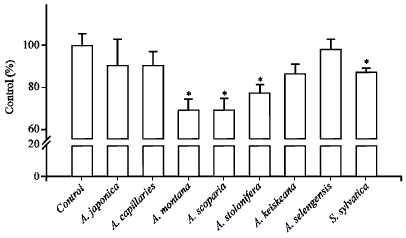 | |
| Fig. 4: | Nitric oxide radical scavenging activity of Artemisia sp. plants-extract, *p<0.05 vs. control (treated only with sodium nitroprusside, 20 mM) |
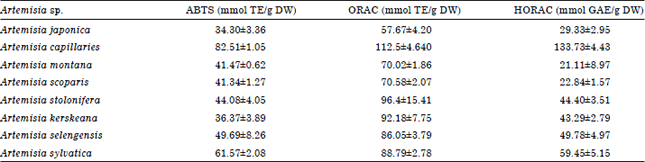
| Table 1: | ABTS, ORAC and HORAC capacity of Artemisia sp. plants |
 | |
The quenching NO radical by plant extracts was measured by Griess reaction. As shown in Fig. 4, A. montana and A. scoparia (300 μg mL¯1) showed approximately 30.7 and 30.7% of NO radical (10 mM) scavenging activity. And other Artemisia sp. plant extracts showed approximately 1.9-20% of NO radical quenching and were appeared in a concentration-dependent manner.
ABTS free radical scavenging activity, ORAC and HORAC of Artemisia sp. plants extract: Artemisia sp. plants-extract exhibited free radical scavenging activities for ABTS radical quenching activity, ORAC and HORAC (Table 1). Artemisia sp. plants-extract showed approximately 34.3-82.51 mmol TE/g DW in ABTS scavenging activity. Artemisia japonica and A. capillaries had approximately 34.3 which was the lowest and 82.5 mmol TE/g DW which was the highest, in ABTS quenching activity, respectively.
Peroxyl Radical Scavenging Capacity (ORAC) of Artemisia sp. plants extract had s similar pattern to hydroxyl radical antioxidant capacity (HORAC). Artemisia sp. plants-extract had approximately 57.67-112.5 mmol TE/g DW in ORAC. Artemisia japonica had the smallest ORAC of 57.67 mmol TE/g DW and A. capillaries had approximately 1.95 times more (112.5 mmol TE/g DW) than A. japonica which was the highest ORAC value among eight Artemisia sp. plants-extract. In addition, the highest value of HORAC was showed in A. capillaries extract.
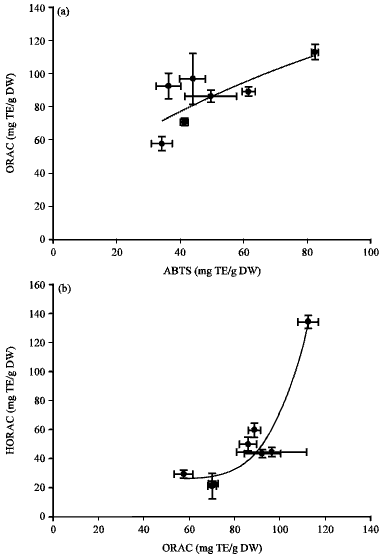 | |
| Fig. 5(a-b): | Correlations between ABTS, ORAC and HORAC of Artemisia sp. plants-extract, (a) ABTS vs. ORAC (R2 = 0.5495) and (b) ORAC vs. HORAC (R2 = 0.9164) |
Artemisia capillaries had approximately 133.73 mmol GAE/g DW in HORAC which was the highest HORAC value in tested Artemisia sp. plants extract and was approximately 6.33 times more than that of A. montana.
Thus A. capillaries had the most potent antioxidant activity in ABTS, ORAC and HORAC value. The potent antioxidant activity of A. capillaries is supposed to be due to the highest content in total phenolic compounds/total flavonoids of A. capillaries. Artemisia capillaries had much larger increase range in antioxidant activity than expected, as compared between the content of phenolic compounds/total flavonoids and antioxidant activity. Therefore, A. capillaries is considered as an effective antioxidant material in foods. In other hand, the content of total phenolic compounds and antioxidant activity did not have the positive linear correlation (R2 = 0.1642). The reason is supposed to have various kinds of organic acid or phenolic compounds without antioxidant activity and to have different amount of solid materials in Artemisia sp. plants-extract. ABTS radical scavenging activity, ORAC and HORAC of Artemisia sp. plants-extract had the positive correlation, as shown in Fig. 5a, b. The ABTS radical scavenging activity and ORAC showed R2 = 0.5495 of correlation and ORAC and HORAC showed R2 = 0.9164 of correlation in eight Artemisia sp. plants-extract. Diverse free radicals are detected by a specific assay method. The ABTS free radical scavenge, ORAC and HORAC of Artemisia sp. plants extract are expected to have a similar trend.
Generally, ABTS method detects the change of ABTS free radical in a relative short reaction time, less than 6 min and actually ABTS free radical is not found in vivo (Perez-Jimenez et al., 2008). The ORAC method can detect the reaction kinetics varied by concentration of antioxidant and scavenging times. And, ORAC assay evaluates antioxidative activity for peroxyl radicals in vivo but some protein may interfere the effect of ORAC value (Perez-Jimenez et al., 2008). The ORAC and HORAC method are accepted and used for the evaluation of antioxidant activity for food materials, pharmaceuticals and plant extracts and are reported to be more sensitive than other antioxidant measuring methods (Ciz et al., 2010).
Artemisia sp. have been used as a traditional medicine in foreign countries and many bioactive materials, such as flavonoids, terpenoids and sesquiterpenoids, were isolated and studied (Skowyra et al., 2014; Singh et al., 2011; Moldovan et al., 2011; Brisibe et al., 2009). It is reported that A. montana extract had approximately 97.16±1.37 mg caffeic acid/g DW and 38.62±1.50 mg quercetin/g DW in total phenolic content and total flavonoid content (Moldovan et al., 2011) which showed the similar result to content of total phenolic compounds/total flavonoids of A. montana in our study. In addition, ORAC of A. montana extract had approximately 700-920 μmol TE/g DW which was less than our result for antioxidant activity of A. montana extract. However, ORAC and TEAC of plant extract showed a linear correlation (R2 = 0.908) (Moldovan et al., 2011). The correlation between the total phenolic acids and ORAC had a linearity with R2 = 0.903 and 0.975, respectively (Moldovan et al., 2011), not being consistent to our result for Artemisia sp. plants extract. A. annua had low contents of total phenolic acids and total flavonoid, being approximately 23.36±0.92 mg GAE/g DW and 2.68±0.07 mg CE/g DW (Skowyra et al., 2014), as compared with our results for Artemisia sp. plant extract. Additionally, ABTS and ORAC of A. annua extract had approximately 314.99±7.70 and 736.26±17.55 μmol TE/g DW which were less than present results.
The purpose of this study is to evaluate the potential antioxidant activity of Artemisia sp. plants, used as a food material and a pharmaceutical in Korea. Artemisia capillaries is investigated to have a potent antioxidant activity in DPPH/ABTS radical quenching activity, ORAC and HORAC. Artemisia capillaries may be expected to provide the antioxidant activity in foods and to act the protective function for cellular oxidative stresses, as a functional food material.