Research Article
Fatty Acid Composition of Black Cumin Oil from Iraq
Department of Pharmacognosy and Pharmacology, College of Pharmacy, Hawler Medical University, Erbil, Iraq
Black cumin oil is a fixed oil obtained from seeds of Nigella sativa L. (Ranunculaceae). Nigella is a small genus of annual herbs found in Southern Europe and Western Asia especially in the Mediterranean region. The spicy seeds from this plant have proclaimed medicinal usage dating back to the ancient Egyptians, Greeks and Romans. Seeds of are angular, dark gray in color and are locally known as Habbatus sauda. The seeds are considered carminative, stimulant, diuretic, emmenagogue, galactagogue, whereas its oil is applied externally for skin eruptions (Kritikar and Basu, 2000). The oil is classified as Generally Regarded as Safe (GRAS) by Food and Drug Administration (Burdock, 1997). Many medicinal properties such as bronchodilatory, hypotensive, antibacterial, antifungal, analgesic, anti-inflammatory and immunopotentiating are attributed to its seeds. The chemical composition and medicinal properties of N. sativa have been extensively reviewed (Khan, 1999). In Egypt and the Middle East the black cumin oil is popularly used for certain cases of chronic cough and bronchial asthma. The seed oil has been reported to possess antitumor (Worthen et al., 1998), antioxidant (Burits and Bucar, 2000) antibacterial (Morsi, 2000; Nair et al., 2005), anti-inflammatory (Houghton et al., 1995), hypoglycemic (Al-Hader et al., 1993), CNS depressant (Al-Naggar et al., 2003), antioxidant and immunostimulatory activities (Salem and Hossain, 2000). These activities have been attributed to the fixed oil, volatile oil or their components. As the part of our investigation on the compositions of fixed oils (Kaskoos et al., 2009) and high use of black cumin in Iraqi diet and traditional medicine, it was thought worthwhile to study composition of black cumin oil. The fact that there are few reports of analysis of black cumin oil from Iraq in comparison to other parts of the world also lured us to examine chemically. The aim of this study was to describe the detailed fatty acid composition along with stability and nutritional characteristics of the oil.
MATERIALS AND METHODS
Plant material: Black cumin seeds were procured in May 2009 from local crude drug market in Mosul, Iraq. The plant material was identified and authenticated by comparison with a standard specimen. A voucher specimen (No. BCS/11/09) was retained for further reference.
Methods
Extraction of fixed oil: Black cumin seeds were cleaned, milled and then passed through a 35 mm (42 mesh) sieve. The ground seeds (50 g) were extracted with hexane for 4 h in a Soxhlet apparatus. The extract was concentrated under reduced pressure. The extracted oil was stored at 4°C in the dark. For analysis fatty acids of the extracted oil were esterified with 2 M KOH in MeOH at room temperature as described by AOAC (1990).
GC-FID analysis: The GC analysis of black cumin oil was performed on Perkin-Elmer Clarus 500 equipped with auto-sampler using Supelcowax 10 column (30 mx0.25 mm; film thickness 0.25 μm). The carrier gas used was hydrogen at 10 psi flow pressure; oven temperature was programmed from 130°C, held for 5 min and raised at 4°C min-1 to a final temperature of 240°C and held for 12.5 min. The injector temperature was 260°C and injection volume was 1.5 μL. Detector used was Flame Ionization Detector (FID) and detector temperature was 290°C.
Identification of fatty acids: Most of the fatty acid methyl esters were identified by GC-FID by comparison of their retention times with those of reference standard available in the laboratory and analyzed under same conditions. The fatty acid composition was expressed as percentage of total fatty acid methyl ester in the oil. Peroxidizability Index (PI) was calculated according to equation of Song et al. (2000) as given below:
The solvent extraction of black cumin seeds (50 g) yielded 14.1 mL of crude oil (28.2%, on dry weight basis). Oil had a golden yellow and a strong aromatic odour. The GC-FID analysis resulted in the identification of twenty-six fatty acids in the oil (Fig. 1), which represented 95% of total fatty acid composition (Table 1). The oil consisted of eight saturated fatty acids (15.13%) and eighteen unsaturated fatty acids (79.87%). Linoleic acid (42.76%), oleic acid (16.59%), palmitic acid (8.51%), eicosatrienoic acid (4.71%), eicosapentaenoic acid EPA (5.98%) and docosahexaenoic acid DHA (2.97%) were the major components. The fatty acid composition is similar to earlier reports (Nergiz and Otles, 1993; Nickavar et al., 2003). These reports suggested that the oils of black cumin varieties contained oleic and linoleic acids (18.9-25.0 and 47.5-60.8%, respectively) that is in good agreement with the present study (16.59 and 42.76%, respectively). The ratios of linoleic to oleic acid and unsaturated to saturated fatty acids were found to be 2.57 and 5.27, respectively and are higher than the literature values (Atta, 2003; Ramadan and Moersel, 2003). Peroxidizability index calculated for the oil was 118.27%. It could be predicted that crude oil extracted by solvent extraction is stable to auto-oxidation and rancidity.
 | |
| Fig. 1: | GC-FID chromatogram of black cumin oil (Nigella sativa L.) from Iraq |
| Table 1: | Fatty acid composition of the black cumin (Nigella sativa L.) oil from Iraq |
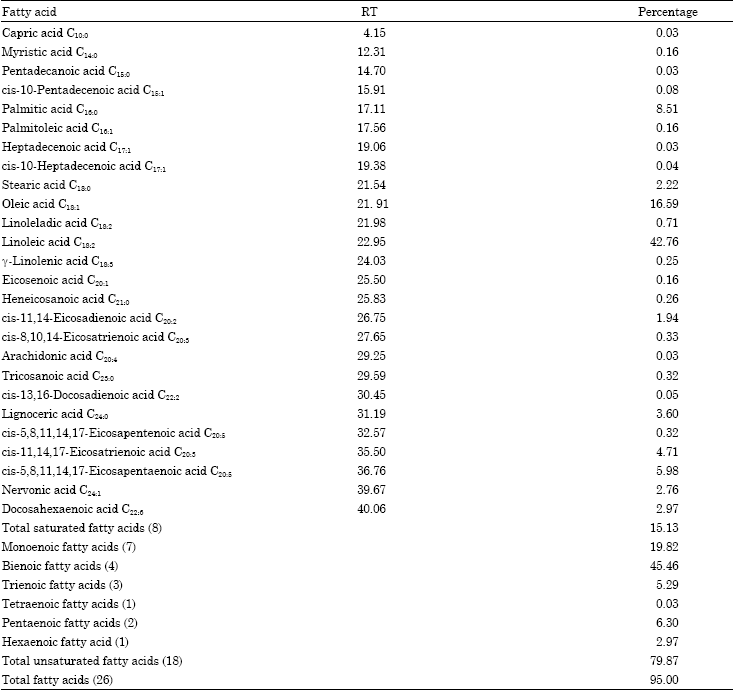 | |
| Compounds listed in order of elution, RT: Retention time, Unsaturated/saturated ratio: 5.27, Peroxidizability Index (PI): 118.27% | |
However, present study reports the presence of eicosapentaenoic acid (20:5n-3; EPA) and docosahexaenoic acid (22:6n-3; DHA) in black cumin oil for the first time (5.98 and 2.97%, respectively). The DHA along with EPA is the predominant n-3 polyunsaturated fatty acid (PUFA) in fish oils. Consumption of fish oils is particularly associated with a low incidence of atherosclerosis and cardiovascular diseases and this prophylactic effect is attributed to n-3 PUFAs, such as EPA and DHA (Sekine et al., 2007). These are highly valued omega-3 fatty acid and are a part of several health foods and nutraceutical preparations. The role of DHA for the growth and functional development of the brain in infants and adults is well established. The inclusion of DHA in the diet improves learning ability, whereas deficiencies of DHA are associated with deficits in learning. The DHA has a positive effect on diseases such as hypertension, arthritis, atherosclerosis, depression, adult-onset diabetes mellitus, myocardial infarction, thrombosis and some cancers (Horrocks and Yeo, 1999; Song et al., 2000).
From these results it may be concluded that the black cumin oil from Iraq has high unsaturated fatty acid content including DHA and can be expected to offer considerable health benefits on consumption and resistance to oxidative rancidity on storage. The source of variability may be related to cultivar or variety, quality, oil processing and accuracy of quantification technique.
The author is grateful Arbro Pharmaceuticals Ltd., Delhi, India, for recording the GC-FID.