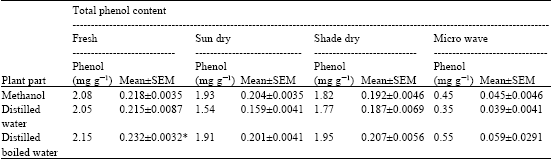
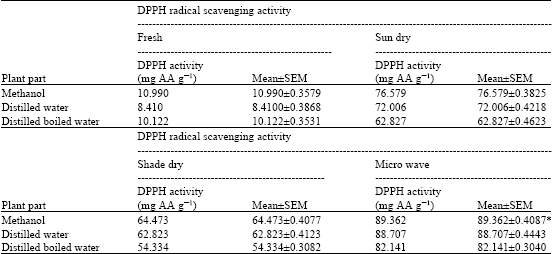
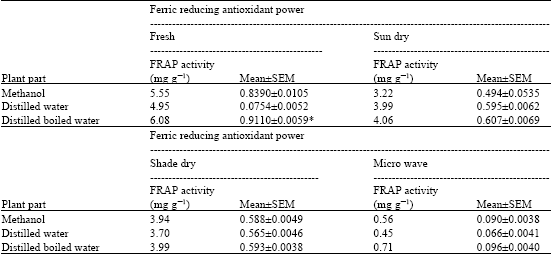
Research Article
Effect of Drying Treatment on the Content of Antioxidants in Enicostemma littorale Blume
Phytomatics Laboratory, Department of Bioinformatics, Bharathiar University, Coimbatore, Tamil Nadu, India
P.T.V. Lakshmi
Phytomatics Laboratory, Department of Bioinformatics, Bharathiar University, Coimbatore, Tamil Nadu, India
A. Annamalai
Department of Biotechnology, Karunya University, Coimbatore, Tamil Nadu, India