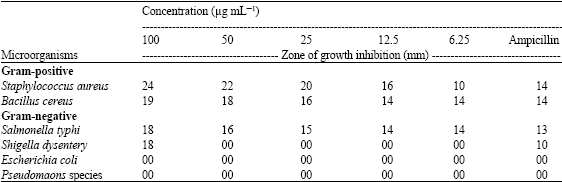
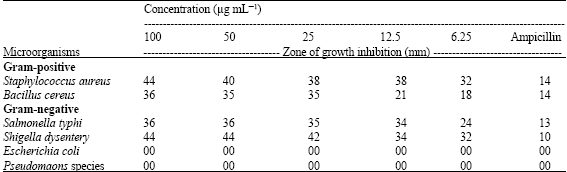
Research Article
Antimicrobial Properties of Osmanthus fragrans (Lour)
Sardar Bhagwan Singh (PG) Institute of Biomedical Sciences and Research, Balawala, Dehradoon (UA), India
Deepak Ganjewala
School of Biotechnology, Chemical and Biomedical Engineering, VII-University, Vellore-632 014 (Tamil Nadu), India