ABSTRACT
The present study was conducted to convert linoleic acid into conjugated linoleic acid by microorganism L. acidophilus MTCC 1473 in vitro and then assess the modulation of functions of various immunocytes viz., T and B lymphocytes and macrophage by bioconverted conjugated linoleic acid in vivo in Swiss albino mice employing various immunological parameters like Development of anti-BSA antibody and anti-SRBC antibody by ELISA and Haemagglutination test (HA) test respectively. Functions of macrophages were checked by using Nitroblue tetrazolium chloride reduction test (NBT), inducible nitric oxide synthetase test (iNOS) Phagocytosis Foot-pad swelling method was employed to assess the T-cells mediated immune response. Results revealed that CLA treatment potentiates the functions of all types of immunocytes i.e., T-cells, B-cells and macrophage.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/rji.2008.45.50
URL: https://scialert.net/abstract/?doi=rji.2008.45.50
INTRODUCTION
Conjugated linoleic acid (CLA) is a term that refers to a mixture of positional and geometric isomers of linoleic acid (LA) in which double bonds are conjugated. CLA has received great attention for their beneficial health properties. It has been reported to prevent carcinogenesis (Ha et al., 1999), atherosclerosis (Nicolosi et al., 1997; Yanwen and Jones, 2004) modulate immune response (Hayek et al., 1999) and reduce body fat (Antonius, 2004; Desroches and Lamarache, 2004). It has been found to possess antioxidative effect which is helpful in preventing many chronic diseases by inhibiting free radicals (Liangli, 2001) within the milk there are different types of fat and fatty acids. The major fatty acids in milk fat range from 4 to 20 carbon chain length. Linoleic acid, an essential dietary fatty acid, contains 18 carbons (C) with two double bonds (C18:2). CLA is a term for specific isomers (forms) of linoleic acid with conjugated double bonds (double bonds adjacent to each other C=C-C=C). Of the 20 different isomers of CLA that have been identified, the cis 9-trans 11 form (commonly called rumenic acid) is believed to be the most common natural form of CLA that is found in diet food and dairy substances and is a major dietary source of CLA in humans (Gillis et al., 2004; Pariza, 2004). The dairy food has been reported to be the most potent source of CLA this lead to the concept of its production from microorganism as it is converted from fatty acids by ruminant bacteria`s. The production of CLA by micro-organism has been reported to convert linoleic acid to Conjugated linoleic acid. Jiang et al. (1998) reported the formation of CLA from linoleic acid by Propinobactrium, Ogawa et al. (2001) and Alonso et al. (2003) reported the formation of CLA from linoleic acid by using Lactobacillus acidophilus bacteria. Keeping above in view the present study was designed to study the effect of microbially transformed CLA as modulator of functions of various immunocytes viz., lymphocytes T and B and Macrophage.
MATERIALS AND METHODS
Microorganisms
Lactobacillus acidophilus MTCC 1473 was procured from Institute of Microbial and Technology, Chandigarh as freeze dried ampoule and revived on specified media and then in MRS medium at 37°C for 24 h and checked for specificity by routine microbiological methods.
Linoleic Acid and Conjugated Linoleic Acid
Linoleic acid and conjugated linoleic acid were purchased from Sigma-Aldrich private limited.
Production of Bioconverted CLA
The Bioconverted CLA was produced by the method of Alonso et al. (2003) by using Linoleic acid. Lactobacillus acidophilus was incubated at 37°C for 24, 48 h, respectively in the presence of different concentrations of linoleic acid. Thereafter flasks were centrifuged and supernatant was collected. The supernatant was analyzed by TLC (Thin layer chromatography). The supernatant was mixed with internal standard heptadecanoic acid and again centrifuged. The whole sample was collected in round bottom flask and dried under Nitrogen gas at 65°C hot temperature in LV-rotary evaporator. The methylation of mixture was done by adding 14% of Boron- Trifluoride in methanol. The organic layer was recovered and analyzed for CLA production by using Gas-chromatography (National institute of Pharmaceutical Education and Research (NIPER, Mohali) CLA peaks were identified with Retention time (RT) and were compared with commercial CLA (Sigma-Aldrich). This Bioconverted CLA was used in mice to study its immunomodulatory Potential.
Immunization
1% BSA in 200 μL per animal was given i.p. as a source of antigen in 3 doses at weekly intervals. In addition the animals were immunized with single dose of sheep red blood cells four days prior to sacrifice.
Animals
Specific pathogen free Swiss-albino mice aged 6 to 8 weeks were purchased from Haryana Agriculture University, India. All experiments were employed in accordance with Institutional Ethics Committee (IEC).
Groups of Animals
Animals were divided into two groups, Test and control (CNT) groups (n = 15 each) Test group animals were given Bioconverted CLA at the rate of 1 mg kg-1 body weight for 7 days Intraperitonially and also normal diet control group (CNT) of animals were given normal diet.
Follow up of the Study
The blood samples of the animals were collected on 15th day from retro-orbit plexus and centrifuged to separate the serum for humoral immune response. After that the mice were anaesthetized and sacrificed. Their spleen was collected for checking the immune response.
Immunological Parameters
Following Immunological tests were employed to study the immune response:
Nitro Blue Tetrazolium Reduction Test (NBT)
The NBT reduction test was employed to assess the function of phagocytes in collected splenocytes as described by Hudson and Hay (1989). Splenocyte suspension from each mouse was incubated in the presence (test) or absence of NBT and formazon formed was extracted in dioxan. The reduction in NBT was measured spectrophotometrically at 520 nm using UV-Vis spectrophotometer, against dioxan as standard:
iNOS (Inducible Nitric Oxide Synthase Test)
The iNOS activity was measured using the method of Stuehr and Marletta (1987)using arginine and Greiss reagent. The purple colour developed was noted andabsorbance was determined with a UV-Vis spectrophotometer (Shimadzu) at 540 nm against control.
Phagocytosis
Phagocytosis test was carried out by the method given by Reghuramulu et al. (1983). The splenocytes and bacterial suspension were mixed in 1:2 ratio and incubated at 37°C for 60 min. Then added 100 μL sterile distilled water to above suspension (to lyse the splenocytes). 100 μL of above suspension was spread on nutrient agar plates using a spreader. The plates were incubated at 37°C for 24 h and the colonies were counted the next day. Plated bacterial suspension was used as control. Phagocytosis (%) was calculated as follows:
Delayed Type of Hypersensitivity (DTH)
The delayed type of hypersensitivity a measure of the CMI was assessed by the foot pad swelling method given by Hudson and Hay (1989). The swelling in foot pad was noticed after 24-48 h in both the foot pads after carrigenean induced in the right hind foot Pad and normal saline in the left hind foot- pad. The swelling in the foot was measured with micro vernier caliper at 0, 24 and 48 h after the challenge. The difference in paw thickness as compared to control was taken as a measure of delayed type of hypersensitivity.
Haemagglutination
Development of Anti-SRBC antibody was checked by Direct Haemagglutination Method given by Hudson and Hay (1989). The titer was checked for antibody by visualizing the plates for mat formation (positive agglutination) and button formation (negative agglutination).
ELISA
Development of the anti BSA albumin antibodies was observed by ELISA test, as given by Hudson and Hay (1989). The absorbance was measured with ELISA reader (BIORAD) at 492 nm.
Statistical Analysis
All the results were expressed as Mean±SEM. Data of tests were statistically analyzed using one-way ANOVA followed by Tukey`s multiple range test, applied for post-hoc analysis. A value of p<0.05 was considered to be statistically significant.
RESULTS
The percentage NBT reduction was checked in test and control group. It was found to be 2.3 times higher in test group as compared to untreated control group statistically significant at p<0.05 (Table 1). Phagocytosis was 2.06 times higher in test group as compared to untreated control group and was statistically significant at p<0.05 (Table 1).
| Table 1: | Influence of CLA on, NBT reduction, iNOS activity and Phagocytosis |
 | |
The results are presented as Mean±SEM (n = 10) p-values, significantly different from the control: *<0.05 | |
| Table 2: | The delayed type of hypersensitivity responses among animal groups |
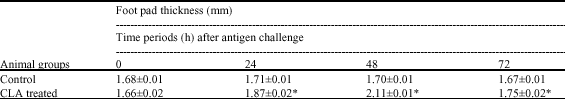 | |
The results are presented as Mean±SEM (n = 10) p-values, significantly different from the control: *<0.05 | |
| Table 3: | Development of Anti- SRBC antibody titer in animal groups |
| Table 4: | Development of Anti- BSA antibody titer in animal groups |
The iNOS activity was 3.0 times higher in test group as compared to untreated control group and was statistically significant at p<0.05 (Table 1).
The DTH response given in Table 2 was found to be maximally effective at 48 h and it was found that CLA enhanced it by 1.1 times in test as compared to control group which was statistically significant at p<0.05 (Table 2).
The humoral immune response i.e., the function of T cells dependent B cells response was assessed by Haemagglutination showed that in CLA animals anti-SRBC antibody titer was 2 times higher as compared to control group (Table 3). Similar to anti-SRBC, anti-BSA antibody was also found to be 4 times higher in test than control group (Table 4).
DISCUSSION
The present study was conducted to see the impact of bioconverted conjugated linoleic acid (CLA) on functions of various immunocytes. The results showed that cell mediated as well as humoral immunity and macrophage functions were potentiated in animals treated with Bioconverted Conjugated linoleic acid as was observed by NBT, INO`S , phagocytosis, DTH, anti-SRBC antibody as well as anti-BSA antibody. Present results corroborate the earlier findings by Yang et al. (2003), Hayek et al. (1999), Yamasaki and Chujo (2003) and Loscher (2005). Yang and Cook (2003), whoobserved immunopotentiation and showed this potentiation could be due to decrease in TNF- factor and production of cytokines. Hayek et al. (1999) observed CLA influences the immune response in mice. Yamasaki (2003) observed production of immunoglobulin in mice by Conjugated linoleic acid. Loscher (2005) showed that conjugated linoleic acid suppresses NF-kB activation and IL-12 production in dendritic cells through ERK-mediated IL-10 induction. From this study as well as the previous reports discussed here show that CLA potentiates the function of all types of immunocytes i.e., T cells, as revealed by DTH results, B-cells as observed by raised antibody titers in CLA treated animals as well as macrophage functions. as was clear by the higher NBT reduction, iNOS as well as Phagocytosis in CLA treated animals as compared to control group animals.
CONCLUSION
In the present study, CLA was produced by bioconversion of linoleic acid in vitro to conjugated linoleic acid by Lactobacillus acidophilus. On the other hand linoleic acid has been reported in many food articles. Hence many food items can be used for the production of CLA in vitro by Lactobacillus acidophilus and collected CLA may be applied to prepare neutraceuticals or Lactobacillus acidophilus or probiotics can be given along with foods and linoleic acid so that bioconversion occur in body itself. CLA can be applied as an alternative source of immunomodulator or immunotherapeutic agent in various infected or non-infected immune related diseases.
ACKNOWLEDGMENT
The financial support to the Department by DST, Government. of India, under the FIST Programme under which the instruments were purchased by the department is fully acknowledge (Grant No. SR/FST/LSI-091/2002, Dated 3.9.2003)
REFERENCES
- Alonso, L., E P. Custea and S.E. Gilliland, 2003. Production of free conjugated linoleic acid by Lactobacillus acidophilus and Lactobacillus casei of Human intestinal Origin. J. Diary Sci., 86: 1941-1946.
PubMed - Antonius, H.M., 2004. Effect of conjugated linoleic acid on body composition and plasma lipids in humans. Am. J. Clin. Nutr., 79: 352-361.
PubMed - Loscher, C.E., 2005. Conjugated linoleic acid suppresses NF (Kappa) B activation and IL-12 Production in Dendritic cells through ERK-mediated IL-10 induction. J. Immunol., 175: 4990-4998.
PubMed - Sophie, D. and L. Benoit, 2004. Metabolic syndrome and effect of conjugated linoleic acid on obesity and lipoprotein disorders. Am. J. Clin. Nutr., 79: 1149S-1152S.
PubMed - Gillis, M.H., S.K. Duckett and J.R. Sackmann, 2004. Effect of supplemental rumen protected conjugated linoleic acid on feedlot performance, carcass quality and leptin concentration in beef cattle. J. Anim. Sci., 82: 851-859.
PubMed - Hayek, M.G., S.N. Han, D. Wu, B.A. Watkins and M. Meydani et al., 1999. Dietary conjugated linoleic acid influences the immune response of young and old C57BL/6NCrlBR mice. J. Nutr., 129: 32-38.
PubMed - Ip, C., S.F. Chin, J.A. Scimeca and M.W. Pariza, 1991. Mammary cancer prevention by conjugated dienoic derivative of linoleic acid. Cancer Res., 51: 6118-6124.
PubMed - Jiang, J., L. Bjorck and R. Fonden, 1998. Production of conjugated linoleic acid by diary starter culture. J. Applied Microbiol., 85: 95-102.
PubMed - Liangli, L.Y., D. Adams and M. Gabel, 2001. Free radical scavenging properties of conjugated linoleic acid. J. Agric. Food. Chem., 49: 3452-3456.
CrossRef - Liangli, L.Y., D. Adams and M. Gabel, 2002. Conjugated linoleic acid isomer differ in their free radical scavenging properties. J. Agric. Food. Chem., 50: 4135-4140.
Direct Link - Nicolosi, R.J., E.J. Rogers, D. Kritchevsky, J.A. Scimeca and P.J. Huth, 1997. Dietary conjugated linoleic acid reduces plasma lipoproteins and early aortic atherosclerosis in hypercholesterolemic hamsters. Artery, 22: 266-277.
PubMed - Ogawa, J., K. Matsumura, S. Kishino, Y. Omura and S. Shimizu, 2001. Conjugated linoleic acid accumulation via 10-hydroxy-12-octadecaenoic acid during microaerobic transformation of linoleic acid by Lactobacillus acidophilus. Applied Environ. Microbiol., 67: 1246-1252.
CrossRef - Pariza, M.W., 2004. Perspective on the safety on the safety and effectiveness of conjugated linoleic acid. Am. J. Clin. Nutr., 79: 1132S-1136S.
PubMed - Yanwen, W. and P.J.H. Jones, 2004. Dietary conjugated linoleic acid and body composition. Am. J. Clin. Nutr., 79: 1153S-1158S.
PubMed - Yang, M., M.E. Cook and M.W. Pariza, 2003. Dietary conjugated linoleic acid decreased cachexia, macrophage and production of cytokines. Exp. Biol. Med., 228: 51-58.
PubMedDirect Link - Yamasaki, M. and H. Chujo, 2003. Immunoglobulin and cytokine production from spleen lymphocytes is modulated in C57BL/6J mice by dietary cis-9, trans-11 and trans-10, cis-12 conjugated linoleic acid. J. Nutr., 133: 784-788.
PubMed








