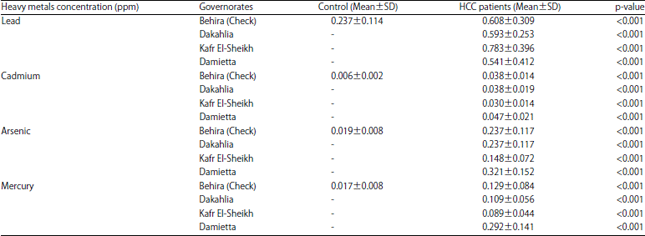
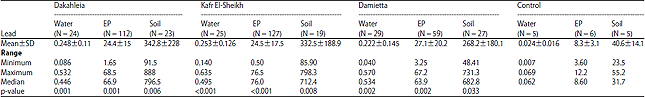
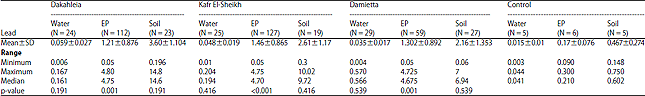
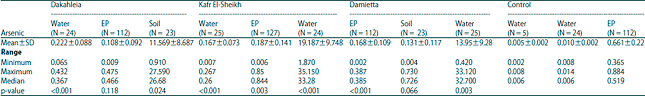
Research Article
Hepatocellular Carcinoma in Populations Consuming Heavy Metals Contaminated Plants in North Delta, Egypt
Departmant of Plant Pathology, Faculty of Agriculture, Mansoura University, 35516 Mansoura, Egypt
LiveDNA: 20.587
Ekbal M. Abo-Hashem
Departmant of Clinical Pathology, Faculty of Medicine, Mansoura University, Mansoura, Egypt
Yasser M. Shabana
Departmant of Plant Pathology, Faculty of Agriculture, Mansoura University, 35516 Mansoura, Egypt
Mohamed A. El-Metwally
Departmant of Mycology Research, Plant Pathology Research Institute, Agricultural Research Center, Giza, Egypt
LiveDNA: 20.359
Ghada El-Kannishy
Departmant of Internal Medicine, Faculty of Medicine, Mansoura University, Mansoura, Egypt
Ali M. El-Adl
Departmant of Genetic, Faculty of Agriculture, Mansoura University, 35516 Mansoura, Egypt
Rokiah Anwar
Departmant of Internal Medicine, Faculty of Medicine, Mansoura University, Mansoura, Egypt
Eman Fawzy
Departmant of Clinical Pathology, Mansoura Fiver Hospital, Mansoura, Egypt
Narmin Saied
Departmant of Clinical Pathology, Faculty of Medicine, Mansoura University, Mansoura, Egypt
Mustafa M. El-Zayat
Genetic Engineering and Biotechnology Unit, Mansoura University, 35516 Mansoura, Egypt