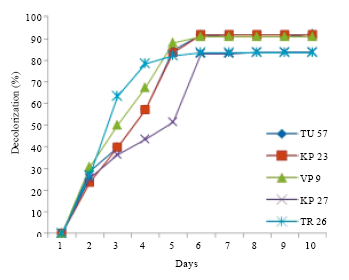
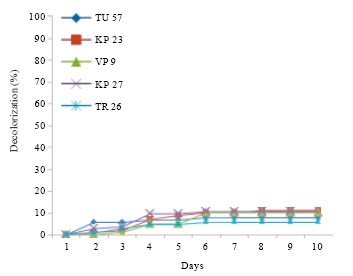
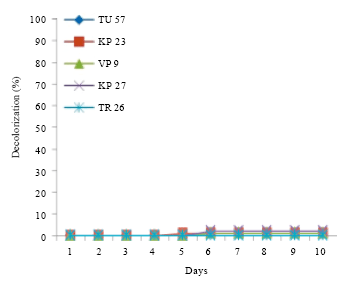
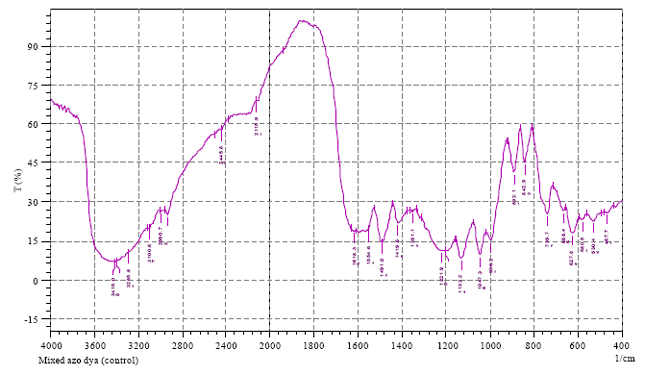
Research Article
Biodegradation of Mixed Textile Dyes by Bacterial Strains Isolated from Dyewaste Effluent
Research and Development Centre, Bharathiar University, Coimbatore, India
R. Subashkumar
PG and Research Department of Biotechnology, Kongunadu Arts and Science College, Coimbatore, India
K. Vijayaraman
Principal Investigator, KSG College of Arts and Science, Coimbatore, India