ABSTRACT
Thiobencarb herbicide toxicity to growth of Protosiphon botryoides (Chlorococcal alga) isolated from an Egyptian paddy was studied using batch culture technique. After sixteen days of incubation in growth medium in presence of 2 ppm thiobencarb, Protosiphon cells count, chlorophyll a content and growth rate were significantly reduced. On the other hand, generation time elevated to be 10.8 day-1 respect to control 6.9 day-1. Electron microscope examination of Protosiphon cells exposed to sublethal concentration (2 ppm) revealed that spores liberation from mother cell wall was delayed; this phenomenon may be attributed to changes in the cell wall structure of the treated cells or physiological reasons induced by the used herbicide or both factors. In conclusion, ultrastructural result implies that reduction in growth of the treated Protosiphon was due to the effect of thiobencarb herbicide high concentration on algal reproduction through retardation on liberation of spores from the mother cell.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/rjet.2010.23.31
URL: https://scialert.net/abstract/?doi=rjet.2010.23.31
INTRODUCTION
Thiobencarb is a thiocarbamate herbicide widely used for weed control in Egyptian paddy fields. It interferes with protein and fatty acids synthesis and inhibits photosynthesis (Corbett et al., 1984; Tomlin, 2003). Beside the use of thiobencarb by direct application for weed control, it may enter to aquatic ecosystem through off site movement from treated fields, by surface run off or adsorbed to suspended particles.
Because of their short generation times, microalgae respond rapidly to environmental changes and thus, may report impacts on higher organisms, which generally respond on longer time scales (McCormick and Cairns, 1994). Furthermore, microalgal tests are generally sensitive, rapid and low-cost effective (Sosak-Swiderska et al., 1998).
There are many studies about the effect of herbicides on algal growth, biochemical composition, metabolic activities and the ultrastructure morphology (Liu and Xiong, 2009; Vendrell et al., 2009; Gonzalez-Barreiro et al., 2006; Rioboo et al., 2002; Kobbia et al., 2001; Caux et al., 1996; El-sheekh et al., 1994). Xia (2005) concluded that, high thiobencarb concentration (10 mg L-1) significantly reduced the biomass yield, protein content and photosynthetic rate in N. sphaeroides colonies. Bhunia et al. (1991) found that thiobencarb at concentrations ranging from 2 to 6 mg L-1 led to a significant decrease in growth, DNA, RNA and total protein content of Nostoc muscorum. Some researchers had been reported that cellular Chl a content was significantly reduced under thiobencarb treatment in Protosiphon botryoides (Eladel et al., 1998) and Anabaena variabilis (Battah et al., 2001).
Since, algae are the primary producers of aquatic ecosystem; the damage to algae may affect both function and structure of the whole ecosystem. Hence, it is of great importance evaluating the impact of thiobencarb to non-target algal organisms. The addition of thiobencarb herbicide to Protosiphon growth medium will certainly bring some changes in its growth. The present study aimed at evaluating the response of Protosiphon botryoides alga to thiobencarb herbicide.
MATERIALS AND METHODS
Organism and Culture Conditions
Protosiphon botryoides (Kutezing) Klebs, a chlorococcal alga, was isolated from paddy field soil samples of Benha, Egypt (2004). Isolation and purification was made by dilution and plating technique. Cultures were grown in 250 mL flasks containing 100 mL of Bold’s basal medium (Bold and Wynne, 1985) and incubated in a controlled growth chamber at 24±2°C and 70 μmol/m2/sec photosynthetically active radiation provided by cool white fluorescent lamps set on 16:8-light:dark photoregime for sixteen days. All cultures were shaken twice daily to prevent cells from clumping.
Treatment with Thiobencarb
Thiobencarb is a thiocarbamate herbicide with moderate water solubility (30 mg L-1 at 20°C). Concentrated herbicide stock prepared from thiobencarb herbicide (technical grade, 95%) by appropriate dilution in acetone. Aliquots of the stock added to each culture to obtain a final concentration of 0, 0.5, 1, 2, 4 and 8 ppm. The carrier solvent acetone completely evaporated to dryness, the culture medium dispensed, equilibrated and the flasks left for one day to obtain aqueous solutions. All cultures (three per treatment) received identical inocula and incubated under the prescribed growth conditions.
Growth Measurements
Protosiphon botryoides algal cells were counted using haemocytometer (Levy and Levy-Hausser corpuscle counting chamber). Chlorophyll a was determined fluorometrically according to APHA (1992).
Algal growth inhibition (I%) respect to the control through cells count (US Environmental Protection Agency, 1989) was calculated as follows:
Where:
| T | = | Number of cells mL-1 in treated cultures |
| C | = | Number of cells mL-1 in control cultures |
Growth rate (μ) and the generation time (GT) of the cultures in the presence and absence of thiobencarb determined by the following equations (Reynolds, 1984):
Where:
| X2 | = | Cells number at end of selected time interval |
| X1 | = | Cells number at beginning of selected time interval |
| T2-T1 | = | Elapsed time per day between selected intervals |
Transmission Electron Microscopy
After sixteen days of treatment with thiobencarb, Protosiphon botryoides cells exposed to sublethal concentration (2 ppm) of thiobencarb and control cells were centrifuged at 1500 rpm for 15 min, washed in thiobencarb-free culture medium and then fixed with 3% glutaraldehyde in phosphate buffered, they were postfixed in 1% osmium tetroxide, dehydrated by series of ethanol and embedded in Araldite resine. Ultrathin sections stained with uranyl acetate and lead citrate and examined using electron microscope (Corradi et al., 1995).
RESULTS
Growth curves of Protosiphon botryoides incubated in growth media containing thiobencarb for sixteen days were clearly different from the control. At 0.5 and 1 ppm Protosiphon growth exceeded control values after 4-6 days of growth till the end of the experiment (Fig. 1). Percent of inhibition, which designated with negative sign plotted down the zero line, replaced with percent of stimulation, which plotted in the positive part over the zero line (Fig. 2). The stimulation percent were 49 and 45% for both 0.5 and 1 ppm, respectively at the end of incubation period.
The growth curves of the cultures exposed to thiobencarb doses above 2 ppm showed a different pattern of growth from control, being more flattened instead of the characteristic exponential curve (Fig. 1). Complete growth inhibition was recorded at 4 and 8 ppm thiobencarb concentrations, where Protosiphon was treated for sixteen days (Fig. 2).
After sixteen days growth, cell number and chlorophyll a content were higher than the control at thiobencarb low concentrations (0.5 and 1 ppm). However, Protosiphon cultures treated with 2, 4 and 8 ppm thiobencarb produced cell number and chlorophyll a content clearly lower than that produced within control (Fig. 3, Table 1).
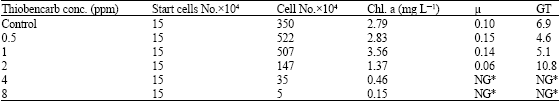
Growth rate (μ) of Protosiphon cultures exposed to 2 ppm dose of thiobencarb was 0.06 day-1, being nearly half of that recorded within the control (0.1 day-1) (Table 1). Growth rate at 2 ppm dose and above were decreased significantly as respect to the control. On the other hand, at 0.5 and 1 ppm growth rate was higher than the control (Fig. 4).
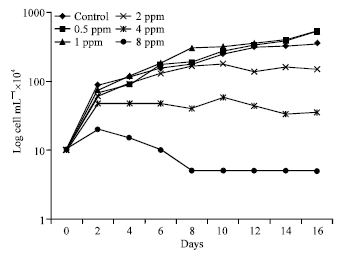 | |
| Fig. 1: | Growth curves of Protosiphon botryoides incubated in different thiobencarb concentrations for 16 days |
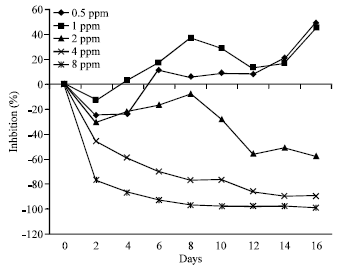 | |
| Fig. 2: | Inhibition % of Protosiphon botryoides incubated in different thiobencarb concentrations for 16 days |
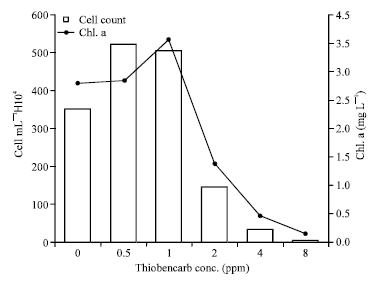 | |
| Fig. 3: | Protosiphon botryoides cell count and chlorophyll a content after incubation in different thiobencarb concentrations for 16 days |
| Table 1: | Protosiphon botryoides cells count, chlorophyll a, growth rate (μ) and Generation Time (GT) after 16 days incubation with different thiobencarb concentrations |
 | |
| NG*: no growth developed | |
After sixteen days Protosiphon generation time curve in (Fig. 4) showed remarkable increase as thiobencarb concentration elevated. At 2 ppm exposed Protosiphon culture, generation time was high (10.8 day-1) as compared with the control 6.9 day-1 (Table 1).
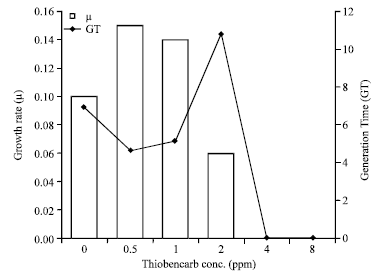 | |
| Fig. 4: | Protosiphon botryoides growth rate (μ) and Generation Time (GT) after incubation in different thiobencarb concentrations for 16 days |
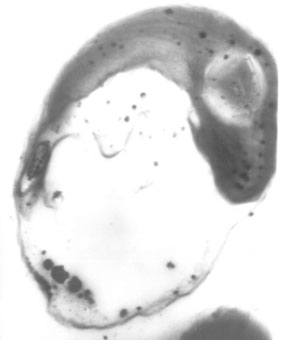 | |
| Fig. 5: | Transmission electron micrograph of Protosiphon botryoides control cell showing a typical structure |
Ultrastructure investigation of Protosiphon cells grown in the used medium free from thiobencarb herbicide (control) by transmission electron microscopy revealed that, control cells maintained their normal characters, the parietal reticulate chloroplast with many scattered pyrenoids and a large vacuole both were surrounded by the cell wall (Fig. 5). Nevertheless, inspection of Fig. 6 one may notice changing in cell wall structure of the treated Protosiphon cells with sublethal concentration (2 ppm) moreover herbicide treated cells undergoing cell division to multispores, which hardly to seen in control cells. The produced spores still enclosed within the treated cells.
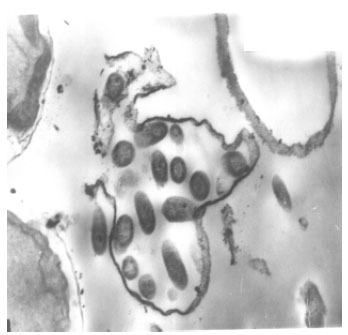 | |
| Fig. 6: | Transmission electron micrograph of Protosiphon botryoides cell exposed to 2 ppm thiobencarb, showing spores still enclosed in the mother cell wall |
The result may reveal that retardation of spores liberation from mother cell wall was one of the modes of action of the used herbicide on Protosiphon alga.
DISCUSSION
Pollution affected algae in different ways. They may be inhibited from growing because of being deprived of sunlight; toxicity of pollutants or may ecologically modify the physical or chemical environment sufficiently to retard or prevent growth.
Thiobencarb low concentrations stimulated Protosiphon growth in terms of increase in cell count and chlorophyll a content. In accordance to the obtained results, stimulation effects are often recorded in algal bioassays with different species and at low concentrations of different pesticides (El-Dib et al., 1991; Haglund, 1997; Franqueira et al., 1999). Other researchers observed the stimulatory phenomenon with lower concentrations of some herbicides (Vendrell et al., 2009; Rioboo et al., 2002; Kobbia et al., 2001; Wong, 2000; Saenz et al., 1993; Aly et al., 1991). It seems that at lower concentration of herbicides, the stimulatory effect may occur because of utilization of the herbicide or its degradation products as an energy source. Other investigator return the stimulation of growth, obtained at the lowest terbutryn concentrations, because the asbility of algal cells and especially of C. vulgaris, to adapt and resist the inhibitory effect of herbicides. This may explain the increase observed in algae in herbicide-treated aquatic ecosystems and perhaps in aquatic ecosystems subjected to toxicants (Shehata et al., 1997).
Because of the effect of high thiobencarb concentrations on Protosiphon growth, the cells needed much time to reproduce themselves, which explain the elevation of generation time value and depression in growth rate value. Growth rate of the chlorophyte Chlorella saccharophila also was inhibited slightly at 3 mg L-1 thiobencarb, but Scenedesmus acutus was inhibited even at 0.01 mg L-1 (Sabater and Carrasco, 1996). Growth pattern, as well as growth rate and generation times exhibited by the cultures of Scenedesmus acutus exposed to 0.05 mg L-1 paraquate and above, were significantly affected (Saenz et al., 1993).
On the bases of the obtained results from electron micrograph, one may attribute the reduction of the growth, growth rate and increasing of the generation time of the treated Protosiphon due to retardation effect of the used herbicide on liberation of spores from the mother cell. This phenomenon seemed to be connected with mechanical reason such as rupture of cell wall, which was not easy to tack place under the used conditions. However, other physiological effects of the herbicide should not be neglected. In accordance to the obtained results, transmission electron microscopy analysis of Chlorella pyrenoidosa cells grown in the presence of two metolachlor herbicides revealed that metolachlor herbicides affected cell morphology, cell division, cell wall, chloroplasts, starch granules and cytoplasm compartments. There were more cells undergoing division that were treated by the two herbicides than the control. This result implies that metolachlor might have some effect on reproduction of this alga (Liu and Xiong, 2009).
Protosiphon zoospores under environmental stress condition may function as isogametes that, upon fusion develop into zygote, which germinate after a period of dormancy into a new Protosiphon (Bold and Wynne, 1985). In agreement with the our results from ultrastructure figures, Corradi et al. (1995) recorded that sublethal concentration of hexavalent chromium induces gametogenesis in Scenedesmus acutus, also Abou-waly (1987) stated that Patron and gardoprim herbicides induces spore formation in Scenedesmus acutus. Cultures exposed to triazine concentrations that affected microalgal growth showed cells larger than normal cells, due to the impossibility of completing their cell division. This effect was reported previously in S. elongatus cultured with atrazine (Gonzalez-Barreiro et al., 2004, 2006). Thus, it was revealed that herbicides have the potential to suppress algal growth, as shown with many algae and force it to respond by spores production.
In conclusion, the present study showed that, the ultrastructural morphology of Protosiphon botryoides cells was affected by thiobencarb herbicide in high concentration.
REFERENCES
- Battah, M.G., E.F. Shabana, I.A. Kobbia and H.M. Eladel, 2001. Differential effects of thiobencarb toxicity on the growth and photosynthesis of Anabaena variabilis with changes in phosphate level. Ecotoxicol. Environ. Saf., 49: 235-239.
CrossRefDirect Link - Bhunia, A.K., N.K. Basu, D. Roy, A. Chakrabarti and S.K. Banerjee, 1991. Growth and chlorophyll a content, nitrogen-fixing ability and certain metabolic activities of Nostoc muscorum: Effect of methylparathion and benthiocarb. Bull. Environ. Contam. Toxicol., 47: 43-50.
CrossRefDirect Link - Corradi, M.G., G. Gorbi and M. Bassi, 1995. Hexavalent chromium induces gametogenesis in the freshwater alga Scenedesmus acutus. Ecotoxicol. Environ. Saf., 30: 106-110.
CrossRef - Eladel, H.M., W.J. Henley and I.A. Kobbia, 1998. Effect of thiobencarb on growth and photosynthesis of the soil alga Protosiphon botryoides (Chlorophyta). J. Applied Phycol., 10: 547-554.
CrossRefDirect Link - El-Dib, M.A., A. Shehata and H.F. Abou-Waly, 1991. Response of freshwater algae (Scenedesmus sp.) to phenylurea herbicides. Water Air Soil Poll., 55: 295-303.
CrossRefDirect Link - El-Sheekh, M.M., H.M. Kotka and H.E. Hammouda, 1994. Effects of atrazine herbicide on growth, photosynthesis, protein synthesis and fatty acid composition in the unicelluar green alga Chlorella kessleri. Ecotoxicol. Environ. Saf., 29: 349-358.
PubMedDirect Link - Franqueira, D., A. Cid, E. Torres, M. Orosa and C. Herrero, 1999. A comparison of the relative sensitivity of structural and functional cellular responses in the alga Chlamydomonas eugametos exposed to the herbicide paraquat. Arch. Environ. Contam. Toxicol., 36: 264-269.
CrossRefDirect Link - Gonzalez-Barreiro, O., C. Rioboo, C. Herrero and A. Cid, 2006. Removal of triazine herbicides from freshwater systems using photosynthetic microorganisms. Environ. Poll., 144: 266-271.
CrossRefDirect Link - Gonzalez-Barreiro, O., C. Rioboo, A. Cid and C. Herrero, 2004. Atrazine induced chlorosis in Synechococcus elongatus cells. Arch. Environ. Contam. Toxicol., 46: 301-307.
CrossRefDirect Link - Kobbia, I.A., M.G. Battah, E.F. Shabana and H.M. Eladel, 2001. Chlorophyll a fluorescence and photosynthetic activity as tools for the evaluation of simazine toxicity to Protosiphon botryoides and Anabaena variabilis. Ecotoxicol. Environ. Saf., 49: 101-105.
CrossRefDirect Link - McCormick, P.V. and J.J. Cairns, 1994. Algae as indicators of environmental change. J. Applied Phycol., 6: 509-526.
CrossRefDirect Link - Rioboo, C., O. Gonzalez, C. Herrero and A. Cid, 2002. Physiological response of freshwater microalga (Chlorella vulgaris) to triazine and phenylurea herbicides. Aquat. Toxicol., 59: 225-235.
Direct Link - Sabater, C. and J.M. Carrasco, 1996. Effect of Thiobencarb on the growth of three species of phytoplankton. Bull. Environ. Contam. Toxicol., 56: 977-984.
CrossRefDirect Link - Saenz, M.E., J. Acorinti and M.D. Tortorelli, 1993. Toxicity of paraquate to a green alga Scenedesmus acutus. J. Environ. Sci. Health, 28: 193-204.
CrossRefDirect Link - Shehata, S.A., M.A. El-Dib and H.F. Abou-Waly, 1997. Effect of certain herbicides on the growth of freshwater algae. Water Air Soil Poll., 100: 1-12.
CrossRefDirect Link - Sosak-Swiderska, B., D. Tyrawska and B. Maslikowska, 1998. Microalgal ecotoxicity test with 3,4- dichloroaniline. Chemosphere, 37: 2975-2982.
CrossRef - Vendrell, E., D.G.B. Ferraz, C. Sabater and J.M. Carrasco, 2009. Effect of glyphosate on growth of four freshwater species of phytoplankton: A microplate bioassay. Bull. Environ. Contam. Toxicol., 82: 538-542.
CrossRefDirect Link - Wong, P., 2000. Effects of 2,4-D glyphosate and paraquat on growth, photosynthesis and chlorophyll-a synthesis of Scenedesmus quadricauda. Chemosphere, 41: 177-182.
CrossRefDirect Link - Xia, J., 2005. Response of growth, photosynthesis and photoinhibition of the edible cyanobacterium Nostoc sphaeroides colonies to thiobencarb herbicide. Chemosphere, 59: 561-566.
CrossRefDirect Link - Liu, H.J. and M. Xiong, 2009. Comparative toxicity of racemic metolachlor and S-metolachlor to Chlorella pyrenoidosa. Aquatic Toxicol., 93: 100-106.
CrossRefDirect Link








