ABSTRACT
The main objective of this investigation was to analyze the spectrum of congenital cardiac malformations, the frequency of extra-cardiac malformations and the proportion of chromosome aberrations among live-born infants in an attempt to generate hypothetical theories of congenital cardiac malformation etiology. The study results showed that out of 1010 Congenital Cardiac Malformation (CCM), 288 were affected by extra cardiac anomalies (28.5%). Atrioventricular Septal Defect (ASD) was found to be the most common CCM associated with ECA (72%) while dextro-transposition of great arteries was the lowest (8.7%). However, syndromes were the commonest extra cardiac anomalies (37%) with congenital cardiac malformation. In conclusion, congenital cardiac malformation were quite common in live-born infants. These defects are often associated with other cardiac and extra cardiac malformations, as well as with chromosome anomalies. Complex heart defects such as hypo-plastic left heart syndrome and double outlet right ventricle were infrequent in live-born infants.
PDF Abstract XML References Citation
How to cite this article
URL: https://scialert.net/abstract/?doi=rjc.2012.12.19
INTRODUCTION
Epidemiological studies conducted over the last 40 years showed that the incidence of congenital heart disease ranged between 4 and 8 per thousand live births (Ferencz et al., 1985; Hoffman and Christianson, 1978; Mitchell et al., 1971). We confirmed higher incidence of congenital cardiac malformations in Saudi population, than most of all incidence figures in medical literature, up to 10.67 per 1000 live birth (Alabdulgader, 2001). Studies of stillborn infants indicated that an incidence of congenital heart disease was ten times greater than that found in live born children (Samanek et al., 1985).
The etiology of most congenital heart defects remains unknown. However, an identifiable cause was found only in approximately 15% of cases (Botto and Correa, 2003). In a group of cases, where causal factors were identified, 10-15% were associated with a gross chromosomal abnormality and around 35% were linked to defects in single genes. By contrast only 2% were linked to environmental factors according to Clark (2001). Congenital heart defects, in cases with identifiable causes, were frequently associated with extra cardiac anomalies.
Ferencz et al. (1989a) carried out infant study in Baltimore-Washington from 1981-1989 (Ferencz et al., 1993) and demonstrated an incidence of 27.71% of extra cardiac anomalies in live-born infants with congenital cardiac malformations defined as malformations of the heart or intrathoracic vessels as compared to only 3.4% in the control population. Previously reported incidence of congenital cardiac malformations with associated extra cardiac anomalies ranged from 9-42% in clinical and 13-37% in autopsy series. In the Baltimore-Washington series, there was a strong association of certain cardiac malformations with extra cardiac anomalies. Atrioventricular Septal Defects (ASD) were associated with extra cardiac anomalies in 51% of cases while Dextro-Transposition of the great arteries was associated with extra cardiac anomalies in only 9% of cases. A study conducted by Losekoot and Becker (1987) confirmed that low incidence of extra cardiac anomalies are associated with Dextro-transposition of the great arteries.
Among the congenital cardiac malformation patients with associated extra cardiac anomalies, the largest sub-group was of those who displayed the features of already defined clinical syndromes. The most common of these was Down’s syndrome (trisomy 21) which was specifically linked to a number of congenital cardiac malformations (Rowe and Uchida, 1961). Other Syndromes have also been linked to specific cardiac malformations. Turner’s syndrome was associated with 75% of cases with left ventricular outflow tract obstruction (Ferencz et al., 1989b) and Noonan’s Syndrome was found in 50% of cases of pulmonary Stenosis (Van der Hauwaert et al., 1978).
In those patients without an identified syndrome, the extra cardiac anomalies occur in the musculoskeletal system in approximately 8.8% of cases, in the central nervous system in 6.9%, the genitor-urinary system in 4.2%, the respiratory system in 3.8% and in less than 1% in the hematological system (Greenwood et al., 1975). Most studies of extra cardiac anomalies associated with congenital cardiovascular malformations are related to western populations.
The Prince Sultan Cardiac Center Al-Hasa is the sole tertiary referral center for all cardiac diseases in this area. The patients in this study represent a sub-group of patients from a comprehensive study of congenital heart disease in the whole Arabian Peninsula. Association between congenital cardiac malformation and extra cardiac anomalies is well documented with broad spectrum which likely reflects complexity of cardiogenesis. The magnitude and type of this association in humans is not known. Worldwide there is a rarity of research on this association. Anticipation of certain associations is highly likely. The main objective of this study was to investigate the association of extra cardiac anomalies with congenital cardiac malformations in a Saudi Arabian population with a known high incidence of congenital heart disease.
MATERIALS AND METHODS
Over a period of 52 months starting in 2004, 1010 children with age ranging from 0-13 years, referred to the Prince Sultan Cardiac Center Al-Hasa. In the study diagnosis of congenital cardiovascular malformation, were considered along with 1010 children (age and sex matched) as controls from the local population. To achieve the objective of this study, Congenital Cardiovascular Malformations (CCM) were defined as any gross structural abnormality of the heart or intrathoracic great vessels which is of actual or potential functional significance. Similarly only Extra Cardiac Anomalies (ECA) of actual or potential clinical significance were included in the analysis and minor anomalies without associated morbidity were excluded. Clinical findings were performed via case-control design (1010 cases+1010 controls), where controls were selected randomly to look for possible specific association of different CCM and ECA which were diagnosed in 1010 cases. Karyotyping and an ultrasound examination were performed.
Full medical history was obtained for each patient and a thorough clinical examination was performed along with electrocardiograms and echocardiography. For those patients with congenital heart disease, further cardiac investigations including cardiac catheterization were performed as a prerequisite to manage the patient.
Routine blood investigation was carried out on all patients including electrophoresis for sickle cell disease, chromosome analysis and routine metabolic screening. Additional investigations were carried out as indicated above to further categorize extra cardiac anomalies, including chest and skeletal X-rays, ultrasound, CT scanning and contrast radiography. Extra cardiac anomalies were categorized into eight groups for analysis i.e., patients with identified chromosomal abnormalities formed one group along with those with identified clinical syndromes. The remaining anomalies were grouped by body system.
Statistical analysis was carried out using a commercially available statistical package (Statistical Analysis System 9.1 for Windows, SAS Institute, Cary, NC) (SAS Institute, 2002). The associations of anomalies were expressed as odds ratios.
RESULTS
A total of 1010 cases with proven congenital cardiac malformations were assessed. The congenital cardiac malformations identified in the study group are summarized in Table 1. Extra-cardiac anomalies were identified in 288 of these cases (28.5%). Extra cardiac anomalies were identified in only 107 (10.8%) of the control group (odds ratio = 3.0, p<0.05). The incidence of type of extra cardiac anomalies in the study and control group is summarized in Table 2.
Furthermore, 107 cases with an identifiable clinical syndrome were identified in the study group in contrast to only 18 in the control group. A total of 88 cases of chromosomal abnormalities were identified in the congenital cardiac malformations group (Table 3). Out of these, 26 were associated with atrioventricular septal defects accounting for 61% of all the cases of atrioventricular septal defects. Down’s syndrome accounted for 81 out of 88 cases with chromosomal abnormalities.
| Table 1: | Congenital cardiac malformations identified |
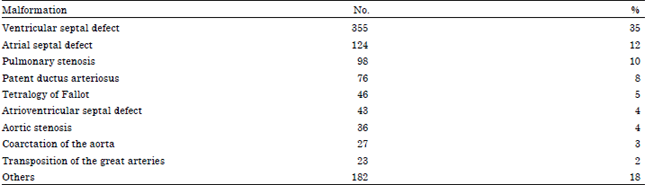 | |
| Table 2: | Associated extra cardiac anomalies |
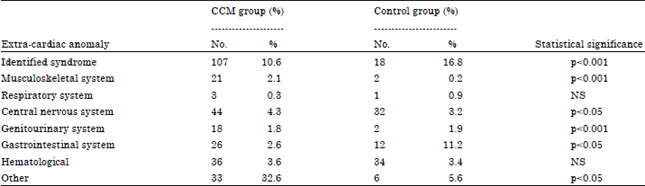 | |
| NS: Not significant | |
| Table 3: | Congenital cardiac malformations associated with chromosomal abnormalities |
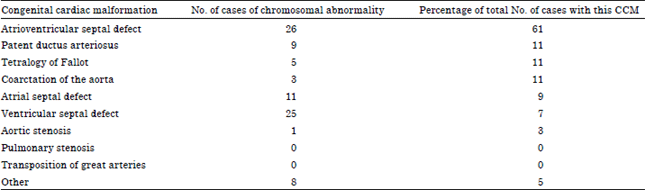 | |
| Table 4: | Chromosomal abnormalities associated with congenital cardiac malformations |
 | |
| Table 5: | Congenital cardiac malformations found in association with Down’s syndrome |
 | |
| Table 6: | Odds ratios calculated between specific congenital cardiac malformations and extra cardiac anomalies |
 | |
The other cases were of Turner’s Syndrome (3), Edward’s Syndrome (2), 4P Tiresome (1) and Unbalanced Translocation 4P Syndrome (1) (Table 4). Down’s Syndrome was associated with atrioventricular septal defects in 32% and ventricular septal defects in 30% of cases. Other associated congenital cardiac malformations included patent ductus arteriosus (11%), atrial septal defects (14%), Tetralogy of Fallot (15%) and others (9%) as summarized in Table 5. When odds ratios were calculated between specific congenital cardiac malformations and extra cardiac anomalies, 4 cardiac malformations were found to be significantly associated with groups of extra cardiac anomalies (Table 6). Nineteen cases of recognized clinical syndromes were also found in association with congenital cardiac malformations and summarized in Table 7. In contrast to the patients with abnormal karyotypes, these cases were associated with transposition of the great arteries (two cases) and pulmonary valve Stenosis or atresia.
| Table 7: | Non-chromosomal syndromes found in association with congenital cardiac malformations |
 | |
DISCUSSION
In the studied population, 28.5% of cases of CCM were associated ECA. This apparently high pick up rate for extra cardiac anomalies may reflect the diligence of the search for extra cardiac anomalies in these cases where many of these were asymptomatic. In contrast, a significantly lower number, 107(10.6%), of extra cardiac anomalies were identified in the control group. The number 28.5% is very similar to 27% incidence of extra cardiac anomalies reported in the Baltimore infant study. Significant associations between congenital cardiac malformations and extra cardiac anomalies were found involving malformations in the central nervous system, genitourinary system gut and musculoskeletal system. Similar findings were reported in the Baltimore study. However, both in our experience and in the Baltimore study, the most common extra cardiac anomalies involved were the central nervous system, the genitourinary system and the gastro intestinal tract (Ferencz et al., 1987). The Baltimore study also observed an increase in abdominal wall abnormalities while present study demonstrated a higher incidence of other musculoskeletal abnormalities. Malformations of the respiratory tract were rare in our series again reflecting the Baltimore experience.
The Baltimore study also showed lack of association between extra cardiac anomalies and Dextro-transposition of the great arteries or left and right sided obstructive lesions. Similarly, the present study identified only two cases of transposition associated with other anomalies. However, one notable feature of this study was the 3.6% incidence of hematological abnormalities in both the study and the control groups which reflected the high incidence of Sickle Cell Disease in the Al-Hasa Region.
Necropsy series have reported much higher incidence of extra cardiac anomalies associated with congenital cardiac malformations. Tennstedt et al. (1999) reported an associated extra cardiac anomalies in 66% of autopsied cases with chromosomal abnormalities. The higher incidence of associated lesions may reflect the additional ability to detect abnormalities at autopsy or a different group of associations in these lethal conditions. However, it is noteworthy that in the present study out of a total of 125 cases, 107 cases (86%) with identifiable clinical syndromes had congenital cardiac anomalies.
As reported in previous studies, the present study found that the commonest extra cardiac anomalies were part of well recognized clinical syndromes many of which have clearly identified chromosomal abnormalities. In this study, Down’s syndrome was present in 81 cases i.e., 8% of all congenital cardiac malformations. Down’s syndrome has long been associated with a number of well recognized cardiac anomalies (Rowe and Uchida, 1961). Our series again confirmed the association with abnormalities of the atrial and ventricular septal and with atrioventricular septal defects, as well as patent ductus arteriosus and Tetralogy of Fallot. Other clinical syndromes are known to have specific associations with particular cardiac anomalies. Turner’s syndrome is frequently associated with left ventricular outflow tract obstruction and Noonan’s syndrome is similarly associated with pulmonary stenosis (Ferencz et al., 1989b; Van der Hauwaert et al., 1978). These common associations were again found in the present study. In necropsy studies, cases of chromosomal abnormalities atrial septal defects, atrioventricular septal defects, ventricular septal defects, double outlet right Ventricle and Hypoplastic Left Heart predominate were found while simple vascular and valvular obstructive lesions and Dextro-transposition were rare. In comparison with our study a higher incidence of chromosomal abnormalities (22%) has been identified prenatally in fetuses with congenital heart defects (Chaoui et al., 1996). Many of these fetuses do not survive to term and thus were reflected in the data collected by pediatric cardiologists (Allan et al., 1991, 1994; Copel et al., 1988). Similarly data from aborted fetuses were not included in studies of the genetic etiology of heart disease (Debrus et al., 1996). This study has demonstrated that the overall pattern of extra-cardiac anomalies found in association with congenital cardiac malformations in the population of Al-Ahsa Region, Eastern Province of Kingdom of Saudi Arabia is generally similar to that reported from Europe and North America. The study again confirmed the strong association of recognized clinical syndromes with specific cardiac abnormalities and of specific cardiac lesions with particular extra cardiac anomalies. Further detailed analysis of the data from this and other studies indicated to examine the relationship between specific extra cardiac anomalies and cardiac anomalies occurring at specific times during gestation. This is especially true where an extra cardiac anomaly is known to be related to a genetic abnormality (chromosomal abnormality or indeed to a known teratogen).
CONCLUSIONS
Association of Congenital Cardiac Malformation with Extra Cardiac Anomalies is well documented. Out of 1010 CCM, 288 (28.5%) were affected with ECA. Atrioventricular septal defect was the most common associated with ECA(72%) while Dextro-Transposition of great arteries is the lowest (8.7%) one. Syndromes are the commonest ECA associated with CCM (37%). Statistically significant association was found between some CCM and certain systems. This is an important evidence to the multiple types of genetic factors in the etiology of CCM. Better knowledge of CCM with ECA and establishment of nationwide registry of CCM (which is at its terminal stage now) should solve some of the mysteries of cardiac dysmorphogenesis in human. In conclusion, CCM, ECA associations and incidence of frequent chromosomal aberrations in Saudi population reflect importance of underlying genetic factors which may have pleiotropic effects and shared common pathways. In this context molecular and bioinformatics approaches uncovering regulatory gene networks may provide insights needed to understand cardiogenesis and Congenital Cardiac Malformation etiology.
ACKNOWLEDGMENTS
The study was supported by a grant from King Abdulaziz City for Science and Technology (KACST) and by a MERIT award to Dr. Abdullah A. Alabdulgader, Principal Investigator. Help and support from Dr Kamal Al-Mubark, Consultant Pediatric Cardiologist during data collection is greatly appreciated. Thanks are extended to Dr Andrew Allen, Head of surgery, PSCCH for revising the manuscript.
REFERENCES
- Allan, L.D., G.K. Sharland, A. Milburn, S.M. Lockhart and A.M.M. Groves et al., 1994. Prospective diagnosis of 1006 consecutive cases of congenital heart disease in the fetus. J. Am. Coll. Cardiol., 23: 1452-1458.
CrossRef - Allan, L.D., G.K. Sharland, S.K. Chita, S. Lockhart and D.J. Maxwell, 1991. Chromosomal anomalies in fetal congenital heart disease. Ultrasound Obstet. Gynecol., 1: 8-11.
PubMed - Botto, L.D. and A. Correa, 2003. Decreasing the burden of congenital heart anomalies: An epidemiologic evaluation of risk factors and survival. Prog. Ped. Cardiol., 18: 111-121.
CrossRef - Copel, J.A., M. Cullen, J.J. Green, M.J. Mahoney, J.C. Hobbins and C.S. Kleinman, 1988. The frequency of aneuploidy in prenatally diagnosed congenital heart disease: An indication for fetal karyotyping. Am. J. Obstet. Gynecol., 158: 409-413.
PubMed - Debrus, S., A. de Meeus, M.K. Jean and P. Bouvagnet, 1996. Genetics of hereditary cardiopathies. Arch. Mal. Coeur. Vaiss., 89: 619-627.
PubMed - Ferencz, C., C.A. Neill, J.A. Boughman, J.D. Rubin, J.I, Brenner and L.W. Perry, 1989. Congenital cardiovascular malformations associated with chromosome abnormalities: An epidemiologic study. J. Pediatr., 114: 79-86.
PubMed - Ferencz, C., J.D. Rubin, R.J. McCarter, J.A. Boughman and P.D. Wilson et al., 1987. Cardiac and noncardiac malformations: Observation in a population-based study. Teratology, 35: 367-378.
PubMed - Ferencz, C., J.D. Rubin, R.J. Mccarter, J.I. Brenner and C.A. Neill et al., 1985. Congenital heart disease: Prevalence at livebirth. The Baltimore-Washington infant study. Am. J. Epi-Demiol., 121: 31-36.
Direct Link - Greenwood, R.D., A. Rosenthal, L. Parisi, D.C. Fyler and A.S. Nadas, 1975. Extracardiac Abnormalities in infants with congenital heart disease. Pediatrics, 55: 485-492.
Direct Link - Hoffman, J.I. and R. Christianson, 1978. Congenital heart disease in a cohort of 19,502 birth with long-term follow-up. Am. J. Cardiol., 42: 641-647.
PubMed - Mitchell, S.C., S.B. Corones and H.W. Berendes, 1971. Congenital heart disease in 56,109 births. Circulation, 43: 323-332.
Direct Link - Rowe, R.D. and I.A. Uchida, 1961. Cardiac malformations in mongolism: A prospective study of 184 mongoloid children. Am. J. Med., 31: 726-735.
CrossRef - Samanek, M., J. Goetzova and D. Benesova, 1985. Distribution of congenital heart malformations in an autopsied child population. Int. J. Cardiol., 8: 235-250.
PubMed - Tennstedt, C., R. Chaoui, H. Korner and M. Dietel, 1999. Spectrum of congenital heart defects and extracardiac malformations associated with chromosomal abnormalities: Results of a seven year necropsy study. Heart, 82: 34-39.
Direct Link - Ferencz, C., J.A. Boughman, C.A. Neill, J.I. Brenner and L.W. Perry, 1989. Congenital cardiovascular malformations: Questions on inheritance. Baltimore Washington Infant Study Group. J. Am. College Cardiol., 14: 756-763.
PubMed - Alabdulgader, A.A., 2001. Congenital heart disease in 740 subjects: Epidemiological aspects. Ann. Trop. Paediatr., 21: 111-118.
PubMed








