ABSTRACT
Studies were conducted to assess if the sludge supplementation in soil induces stress in plants. Tomato plants variety Pusa Hybrid 2 was raised in soils supplemented with sludge in different proportions (25, 50, 75 and 100%). Measurement of morphological, physiological and biochemical indices indicated no alteration in the growth till 50%, though an inhibition in growth was noted at higher concentrations. The levels of malondialdehyde (MDA) and hydrogen peroxide (H2O2) did not show any change in comparison to control till 50% concentration. The increase in MDA and H2O2 at higher concentrations (75 and 100%) suggested the induction of oxidative stress. The accumulation of compatible solutes was evident in plants raised in soil supplemented with high proportions of sludge (75 and 100%). Studies suggested that sludge supplementation in soil promote growth in plants up to a certain concentration without inducing stress.
PDF Abstract XML References Citation
How to cite this article
URL: https://scialert.net/abstract/?doi=rjb.2015.22.29
INTRODUCTION
The treatment of wastewater generates sludge as a waste and its disposal is an issue of major concern. Sludge is organic carbon rich material produced during wastewater treatment. Reuse of sludge in agriculture seems an attractive option as it represents a source of organic matter, nitrogen, phosphorus and other nutrients which improve soil fertility (Garcia et al., 2000; Garcia-Gil et al., 2004; Fernandez et al., 2009). Sludge improves the properties of the soil through organic matter, phosphorus and nitrogen content. The changes in the properties of soil support the growth of plants. Addition of high concentrations of sludge in soil can alter the properties of the soil to a significant extent and hence can act as a stress for plants. Abiotic stress leads to generation of Reactive Oxygen Species (ROS) that cause damage to proteins, lipids, carbohydrates and DNA (Foyer and Noctor, 2000). The H2O2 plays a major role in stress-induced damage and also acts as an integral component of cell signalling cascades (Mittler, 2002; Vranova et al., 2002). It regulates the expression of genes encoding antioxidants, cell rescue/defense proteins and signalling proteins such as kinase, phosphatase and transcription factors (Hung et al., 2005). The oxidative stress caused by ROS initiates the antioxidant defense system in plants. These include enzymatic and non-enzymatic pathways. Besides this, accumulation of compatible solutes such as betaine, proline, polyols, sugars and sugar alcohols is also enhanced during stress. Biosynthesis of compatible solutes also acts as a plants strategy for protection and survival under stress conditions (Chen et al., 2007). Compatible solutes or osmolytes are low molecular weight, highly soluble compounds primarily located in the cytosol and chloroplastic compartments (Bohnert and Shen, 1998; Serraj and Sinclair, 2002). They play a cardinal role in osmoregulation, protection of sub-cellular structures, maintaining plant water status, turgidity and stabilize proteins and cells structures (Yancey et al., 1982). In addition, they also act as small hydrophilic organic molecules, chaperones that help in stabilizing the photosystem II complex, protecting the structure of enzymes and proteins, maintaining membrane integrity and scavenging ROS (Noiraud et al., 2001).
The present studies were undertaken to evaluate if the plants raised in sludge are exposed to any kind of stress. Sludge supplementation alters the soil properties and/or changes the soil matrix. Seedling is considered as the most crucial stage for growth and development of plants therefore the studies were carried out at this stage. The changes in growth were measured using the physiological and biochemical parameters. In addition, another objective of the study was to assess the production of compatible solutes in plants and investigate if they have any role in combating stress (if any).
MATERIALS AND METHODS
Physico-chemical characteristics of sludge and soil: Air-dried sludge was collected from Common Effluent Treatment Plant (CETP) located at Mayapuri, New Delhi, India. The physicochemical parameters such as pH, Electrical Conductivity (EC), Total Dissolved Solutes (TDS), metal content, organic matter, carbon, nitrogen, phosphorus and potassium content were measured. Soil samples taken from the surface at a depth of 10 cm were air dried, homogenized, passed through a 2 mm sieve and analyzed for physico-chemical properties as such as pH, Water Holding Capacity (WHC), Electrical Conductivity (EC), Total Dissolved Solutes (TDS), Organic Matter (OM), available potassium, nitrogen, phosphorus (Dhir and Srivastava, 2013).
Experimental set up: Clay loam garden soil was used for the experiment. The sludge was air-dried, crushed to pass a 2 mm sieve and dry sludge was mixed with soil to have five sludge soil mixtures representing 25, 50, 75 and 100%. The soil without any supplement was referred as the control (C). Ten seeds of tomato were place in pot containing one kilogram of soil-sludge mixtures. Plants were watered every other day. The measurements were done after 30 DAS (Days After Sowing). Plant height and fresh biomass was measured.
Biochemical estimations
Photosynthetic pigments: Fresh leaves (1 g) were extracted with 80% acetone (v/v) and pigment content was estimated spectrophotometrically at 645 and 663 nm and expressed in terms of mg chlorophyll present per gram fresh mass (Hiscox and Israelstam, 1979).
Total soluble protein: Leaves homogenized in Tris-NaOH buffer (pH 7.0) were centrifuged at 12,000 g for 10 min. The supernatant was mixed with Bradford reagent and optical density was measured at 595 nm (Bradford, 1976).
Total soluble sugars: Soluble sugars were quantified following the phenolsulfuric acid method. Five hundred milligram of leaves was extracted in 80% (v/v) methanol heated to 70°C in a water bath. The extract was then centrifuged at 5,000 × g for 10 min. The supernatant was used for the estimation of soluble sugar concentrations. The mixture consisted of 5% phenol and 98% sulphuric acid. The extract was cooled and the absorbance was determined at 490 nm using D-glucose as standard (Dubois et al., 1956).
Malondialdehyde (MDA) levels: The MDA levels were estimated according to the corrected TBA method (Hodges et al., 1999). Two milliliters of extraction solution and 3 mL 0.5% TBA including 5% TCA were mixed vigorously. The mixture was heated at 95°C in a constant temperature water bath for 30 min and then cooled in ice to room temperature. After centrifuging at 5000 × g for 15 min, the supernatant was detected at 450, 532 and 600 nm. The concentration of MDA was determined using the formula CMDA (μmol mL–1) = 6.45×(D532-D600)-0.56×D450, where D450, D532 and D600 are the absorbances at 450, 532 and 600 nm, respectively.
Hydrogen peroxide: Fresh leaves (0.5 g) were frozen in liquid nitrogen and ground to powder in a mortar with pestle, together with 5 mL of 5% TCA and 0.15 g activated charcoal. The mixture was centrifuged at 10,000 × g for 20 min at 4°C. The supernatant was adjusted to pH 8.4 with 17 M ammonia solution and then filtered. The filtrate was divided into aliquots of 1 mL. To one of these, the blank was added 8 μg of catalase and then kept at room temperatures for 10 min. To both aliquots, with and without catalase, 1 mL of colorimetric reagent was added. The reaction solution was incubated for 10 min at 30°C. Absorbance at 505 nm was determined spectrophotometrically. The colorimetric reagent contained 10 mg of 4-aminoantipyrine, 10 mg of phenol, 5 mg of peroxidase (150 U mg–1), dissolved in 50 mL of 100 mM acetic acid buffer (pH 5.6) (Patterson et al., 1984).
Glycine betaine: Glycine betaine was estimated by the method of Grieve and Grattan (1983). Finely ground dried plant tissue (0.5 g) was stirred with 20 mL distilled water for 24 h and filtered. The filtrate was diluted with equal volume of 1 M H2SO4, made into aliquots of 0.5 mL in microcentrifuge tubes, cooled over ice for 1 h and to each of these were added 0.2 mL cold KI-I2 reagent. The reactants were gently stirred, stored at 4°C overnight and centrifuged at 12000 × g for 15 min at 4°C to get the precipitated periodide crystals. The crystals were dissolved in 1, 2-dichloroethane and absorbance was measured at 365 nm after 2 h. Glycine betaine dissolved in 1 M H2SO4 served as standard.
Proline: The plant material was homogenized in 3% aqueous sulfosalicylic acid and the homogenate was centrifuged at 14,000 rpm. The reaction mixture consisted of supernatant, acid ninhydrin and glacial acetic acid. The mixture was boiled at 100°C for 1 h. After termination of reaction in ice bath, the reaction mixture was extracted with toluene and absorbance was read at 520 nm using L-proline as standard (Bates et al., 1973).
Glucose: The 0.5 g of dry leaves was homogenized with 5 mL of 95% ethanol. One milliliter of alcoholic extract was mixed with 3 mL of anthrone (150 mg anthrone and 100 mL of 72% sulphuric acid). The samples were placed in a boiling water bath for 10 min. The light absorption of the samples was estimated at 625 nm by using a spectrophotometer. The soluble sugar contents were determined by using a glucose standard and expressed as mg g–1 DW of leaves (Yemm and Willis, 1954).
Amino acids: Total free amino acids were extracted and estimated by following the method of Moore and Stein (1957). Five hundred milligrams of fresh plant material was homogenized in a mortar and pestle with 80% boiled ethanol. The extract was centrifuged at 800 × g for 15 min and the supernatant was made up to 10 mL with 80% ethanol. In 25 mL test tube, ethanol extract was taken and neutralized with 0.1 N NaOH using the methyl red indicator to which ninhydrin reagent was added. The contents were boiled in a boiling water bath for 20 min and then 5 mL of diluting solution was added, cooled and made up to 25 mL with distilled water. The absorbance was read at 570 nm.
Extraction: The leaves were powdered and 1 g of the powder was extracted continuously with methanol (50 mL). The solution was then swirled for 1 h at room temperature using an orbital shaker. Extracts were then filtered and stored at -20°C for further use.
The total phenolic content was determined using Folin-Ciocalteu reagents with analytical grade gallic acid as the standard. One milliliter of extract was added to deionized water (10 mL) and Folin-Ciocalteu phenol reagent (1 milliliter). After 5 min, 20% sodium carbonate (2.0 mL) was added to the mixture. The mixture was incubated in dark for 1 h. The absorbance was measured at 750 nm using a spectrophotometer. The amount was calculated using gallic acid calibration curve and results were expressed as Gallic Acid Equivalents (GAE) g/g of dry plant matter (Ainsworth and Gillispie, 2007).
Flavonoid content: Flavonoids content was determined with modified aluminum chloride colorimetric method (Woisky and Salatino, 1998). Two hundred fifty milligrams of shoots were extracted with ethanol (20 mL) under reflux in water bath at 80°C for 2 h and filtered. 0.5 mL of ethanol extracts were separately mixed with 1.5 mL of 95% ethanol, 0.1 mL of 10% aluminum chloride, 0.1 mL of 1 M potassium acetate and 2.8 mL of distilled water. After incubation at room temperature for 30 min, the absorbance of the reaction mixture was measured at 415 nm with a Shimadzu UV-160A spectrophotometer (Kyoto, Japan). The final results were calculated as μg g–1 shoot DW. Quercetin was used to make the calibration curve.
Statistical analysis: Mean values from three independent experiments each with three replicates were subjected to Duncan’s Multiple range test to measure level of significance of differences observed between control and treated samples. The rate of photosynthesis was measured using portable PAM fluorometer (mini-PAM, Walz, Effeltrich, Germany) (Dhir and Srivastava, 2013).
RESULTS AND DISCUSSION
Soil analysis: The physical and chemical properties of soil showed an alteration after addition of municipal sludge (Table 1). The water holding capacity, cation exchange capacity and nutrient content were significantly increased after sludge amendment. The soil got enriched with organic matter. The organic carbon and level of nutrients such as nitrogen, phosphorus and potassium were also significantly enhanced (Garcia-Gil et al., 2004; Hussein, 2009).
Plant analysis: Fresh plant biomass showed an enhancement till 50% sludge supplementation, thereafter a decrease was noted. The plant height did show any significant alteration in any of the treatments.
| Table 1: | Changes in the physico-chemical characters of soil after sludge amendment |
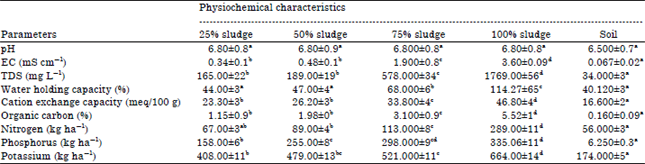 | |
| EC: Electrical conductivity, TDS: Total dissolved solutes, Each value represents Mean±SE, Data followed by different letters in rows are significantly different at p≤0.05 | |
| Table 2: | Morphological and physiological parameters in tomato plants under different treatments |
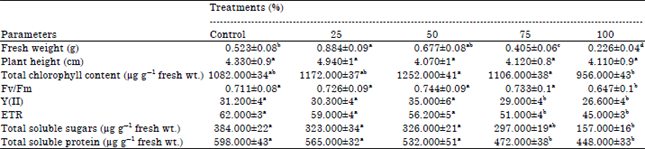 | |
| Each value represents Mean±SE , Data followed by different letters in rows are significantly different at p≤0.05 | |
Sludge supplementation enriches soil with nutrients that support growth and hence increase the biomass production in plants (up to certain proportions). The improvement in the physiological performance also contributes to increase in biomass production. The results are in accordance with previous studies in Zea mays, Corchorus, Eruca, Raphanus, Spinacia and Hordeum vulgare which indicated positive effects of sludge application on plant growth (Mazen, 1995, 2003; Antolin et al., 2005).
Among the biochemical parameters, photosynthetic pigments showed an increase till 75%, thereafter a decline was noted. The enhancement in pigment was significant (1.15 fold enhancement) at 50% sludge treatment. The results are in agreement with previous observations in Corchorus, Daucus, Eruca, Lactuca, Raphanus, Zea mays and Spinacia, wherein plants raised in sludge supplemented soil showed enhanced chlorophyll production (Mazen, 1995, 2003; Mazen et al., 2010). The rate of photosynthesis was not affected till 75%, thereafter a decline was noted (Table 2). The parameters such as Electron Transport Rate (ETR) also showed a significant decline at higher concentrations in comparison to control. Total soluble sugars also showed no alteration till 50% but decline at 75 and 100% in comparison to control. Since, the photosynthetic efficiency (Fv/Fm) was not altered in plants grown in sludge supplemented soils till 50%, therefore soluble sugars also showed no alterations. Sludge-treated plants showed no significant change in total soluble protein content at low concentrations but decline was significant in plants grown at higher concentrations. The alterations in the soluble proteins could be related to change in the activity of enzymes related to nitrate assimilation (Antolin et al., 2010). Total amino acid content, however showed no significant change in the any of the treatments in comparison to control (Table 3).
Malondialdehyde (MDA), one of the decomposition products of polyunsaturated fatty acids of biomembranes is considered as an indicator of oxidative stress in plants. Sludge amendment did not cause increase in MDA at lower concentrations but an increase was noted at higher concentration (Table 3). Hydrogen peroxide, one of the major products of oxidative stress showed a continuous decrease with increase in sludge concentration in soil. The decrease in H2O2 could be related to efficient detoxification machinery acting within the plant to curtail stress (if any). The markers of oxidative stress such as MDA and H2O2 did not depict any significant change in any of the treatments in comparison to control.
Sugars such as sucrose did not show any significant alteration in control and treated samples, while glucose depicted a significant decrease (Table 3). Soluble sugars are supposed to be involved defences and curtailing of ROS (Couee et al., 2006). Proline levels did not show any change till 75%, but a significant increase was noted at 100% sludge treated plants. The accumulation of proline in stressed plants is associated as a strategy adapted by plants to protect damage to the membranes and proteins (Verma, 1999).
| Table 3: | Biochemical studies in tomato plants under different treatments |
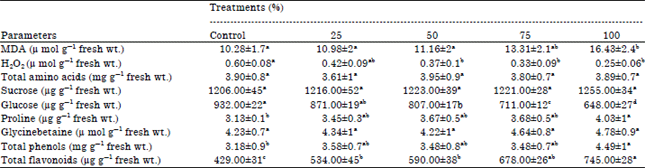 | |
| MDA: Malondialdehyde, Each value represents Mean±SE , Data followed by different letters in rows are significantly different at p≤0.05 | |
Proline has been known to play a role in maintenance of cytoplasmic pH and maintenance of NADP/NADPH ratio (Hare and Cress, 1997). Among other compatible solutes, glycinebetaine and total phenols did not show any significant change in any of the treatments. However, the flavonoids showed a decrease at higher concentrations indicating their role in protection against stress. The flavonoids are supposed to act as antioxidants and protect the plants from physiological and biochemical damage (Pourcel et al., 2007; Ferdinando et al., 2012). It is supposed that sludge supplementation at higher concentrations might be imposing stress in plants, which is being effectively curtailed by compatible solutes such as glucose and flavonoids.
CONCLUSION
The lower concentrations of sludge (up to 50%) do not induce any stress in plants and hence can used as a soil supplement. Sludge amendment at high concentrations might induce oxidative stress as evident by decline in physiological and biochemical parameters. The solutes such as sugars and flavonoids might play an important role in curtailing oxidative stress at higher concentrations.
ACKNOWLEDGMENTS
The financial assistance from University Grants Commission (UGC) to Bhupinder Dhir is gratefully acknowledged. The valuable guidance of mentor, Prof. M.V.Rajam is duly acknowledged.
REFERENCES
- Ainsworth, E.A. and K.M. Gillespie, 2007. Estimation of total phenolic content and other oxidation substrates in plant tissues using Folin-Ciocalteu reagent. Nat. Protocols, 2: 875-877.
CrossRefPubMedDirect Link - Antolin, M.C., I. Muro and M. Sanchez-Diaz, 2010. Sewage sludge application can induce changes in antioxidant status of nodulated alfalfa plants. Ecotoxicol. Environ. Saf., 73: 436-442.
CrossRefDirect Link - Bates, L.S., R.P. Waldren and I.D. Teare, 1973. Rapid determination of free proline for water-stress studies. Plant Soil, 39: 205-207.
CrossRefDirect Link - Bohnert, H.J. and B. Shen, 1998. Transformation and compatible solutes. Scientia Horticulturae, 78: 237-260.
CrossRefDirect Link - Bradford, M.M., 1976. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem., 72: 248-254.
CrossRefPubMedDirect Link - Couee, I., C. Sulmon, G. Gouesbet and A. El Amrani, 2006. Involvement of soluble sugars in reactive oxygen species balance and responses to oxidative stress in plants. J. Exp. Bot., 57: 449-459.
CrossRefDirect Link - Dhir, B. and S. Srivastava, 2013. Supplementation of effluent treated plant biomass in soil and growth/productivity of Triticum aestivum L. and Vigna radiata L. Int. J. Agron. Plant Prod., 4: 3616-3622.
Direct Link - DuBois, M., K.A. Gilles, J.K. Hamilton, P.A. Rebers and F. Smith, 1956. Colorimetric method for determination of sugars and related substances. Anal. Chem., 28: 350-356.
CrossRefDirect Link - Fernandez, J.M., C. Plaza, J.C. Garcia-Gil and A. Polo, 2009. Biochemical properties and barley yield in a semiarid mediterranean soil amended with two kinds of sewage sludge. Applied Soil Ecol., 42: 18-24.
CrossRefDirect Link - Foyer, C.H. and G. Noctor, 2000. Oxygen processing in photosynthesis: Regulation and signalling. New Phytol., 146: 359-388.
CrossRefDirect Link - Garcia, C., T. Hernandez, J.A. Pascual, J.L. Moreno and M. Ros, 2000. Microbial Activity in Soils of SE Spain Exposed to Degradation and Deserification Processes: Strategies for their Rehabilitation. In: Research and Perspectives of Soil Enzymology in Spain, Garcia, C. and T. Hernandez (Eds.). CEBA, Murcia, pp: 93-143.
- Garcia-Gil, J.C., C. Plaza, N. Senesi, G. Brunetti and A. Polo, 2004. Effects of sewage sludge amendment on humic acids and microbiological properties of a semiarid Mediterranean soil. Biol. Fertil. Soils, 39: 320-328.
CrossRefDirect Link - Grieve, C.M. and S.R. Grattan, 1983. Rapid assay for determination of water soluble quaternary ammonium compounds. Plant Soil, 70: 303-307.
CrossRefDirect Link - Hare, P.D. and W.A. Cress, 1997. Metabolic implications of stress-induced proline accumulation in plants. Plant Growth Regul., 21: 79-102.
CrossRefDirect Link - Hiscox, J.D. and G.F. Israelstam, 1979. A method for the extraction of chlorophyll from leaf tissue without maceration. Can. J. Bot., 57: 1332-1334.
CrossRefDirect Link - Hodges, D.M., J.M. DeLong, C.F. Forney and R.K. Prange, 1999. Improving the thiobarbituric acid-reactive-substances assay for estimating lipid peroxidation in plant tissues containing anthocyanin and other interfering compounds. Planta, 207: 604-611.
CrossRefDirect Link - Hussein, A.H.A., 2009. Impact of sewage sludge as organic manure on some soil properties, growth, yield and nutrient contents of cucumber crop. J. Applied Sci., 9: 1401-1411.
CrossRefDirect Link - Hung, S.H., C.W. Yu and C.H. Lin, 2005. Hydrogen peroxide functions as a stress signal in plants. Bot. Bull. Academia Sinica, 64: 1-10.
Direct Link - Pourcel, L., J.M. Routaboul, V. Cheynier, L. Lepiniec and I. Debeaujon, 2007. Flavonoid oxidation in plants: From biochemical properties to physiological functions. Trends Plant Sci., 12: 29-36.
CrossRefDirect Link - Mazen, A.M.A., 1995. Assessment of heavy metal accumulation and performance of some physiological parameters in Zea mays L. and Vicia faba L. grown on soil amended by sewage sludge resulting from sewage water treatment in the State of Qatar. Qatar Univ. Sci. J., 15: 353-359.
Direct Link - Mazen, A., 2003. Heavy metal accumulation and physiological consequences in selected food plants grown on sewage sludge amended soils. Arab Gulf J. Scient. Res., 21: 140-147.
Direct Link - Mazen, A., F.A. Faheed and A.F. Ahmed, 2010. Study of potential impacts of using sewage sludge in the amendment of desert reclaimed soil on wheat and jews mallow plants. Braz. Arch. Biol. Technol., 53: 917-930.
CrossRefDirect Link - Mittler, R., 2002. Oxidative stress, antioxidants and stress tolerance. Trends Plant Sci., 7: 405-410.
CrossRefPubMedDirect Link - Moore, S. and W.H. Stein, 1954. A modified ninhydrin reagent for the photometric determination of amino acids and related compounds. J. Biol. Chem., 211: 907-913.
CrossRefDirect Link - Noiraud, N., L. Maurousset and R. Lemoine, 2001. Transport of polyols in higher plants. Plant Physiol. Biochem., 39: 717-728.
CrossRefDirect Link - Patterson, B.D., E.A. MacRae and I.B. Ferguson, 1984. Estimation of hydrogen peroxide in plant extracts using titanium(IV). Anal. Biochem., 139: 487-492.
CrossRefDirect Link - Serraj, R. and T.R. Sinclair, 2002. Osmolyte accumulation: Can it really help increase crop yield under drought conditions? Plant Cell Environ., 25: 333-341.
CrossRefPubMedDirect Link - Vranova, E., S. Atichartpongkul, R. Villarroel, M. Van Montagu, D. Inze and W. Van Camp, 2002. Comprehensive analysis of gene expression in Nicotiana tabacum leaves acclimated to oxidative stress. Proc. Natl. Acad. Sci. USA., 99: 10870-10875.
CrossRefDirect Link - Yancey, P.H., M.E. Clark, S.C. Hand, R.D. Bowlus and G.N. Somero, 1982. Living with water stress: Evolution of osmolyte systems. Science, 217: 1214-1222.
CrossRefPubMedDirect Link - Yemm, E.W. and A.J. Willis, 1954. The estimation of carbohydrates in plant extracts by anthrone. Biochem. J., 57: 508-514.
PubMedDirect Link - Chen, Z., T.A. Cuin, M. Zhou, A. Twomey, P.N. Bodapati and S.N. Shabala, 2007. Compatible solute accumulation and stress-mitigating effects in barley genotypes contrasting in their salt tolerance. J. Exp. Bot., 58: 4245-4255.
CrossRefDirect Link - Woisky, R.G. and A. Salatino, 1998. Analysis of propolis: Some parameters and procedures for chemical quality control. J. Apicult. Res., 37: 99-105.
CrossRefDirect Link - Antolin, M.C., I. Pascual, C. Garcia, A. Polo and M. Sanchez-Diaz, 2005. Growth, yield and solute content of barley in soils treated with sewage sludge under semiarid Mediterranean conditions. Field Crops Res., 94: 224-237.
CrossRefDirect Link








