Research Article
Sorption of Soil Organic Carbon in Relation to Soil Properties in a Semi-Arid Environment
Department of Soil Science and Technology, Federal University of Technology, P.M.B. 1526, Owerri, Nigeria
INTRODUCTION
Soil organic matter is a complex material consisting of plant biopolymers, animal and microbial residues and humic substances (Chefetz et al., 2002). The amount, chemical composition and polyelectrolytic characteristics of the resulting organic carbon may vary considerably depending on climate, origin and age of the organic matter (Chen et al., 2002).
Soil organic matter affects physical, chemical and biological properties of soils. Soil organic matter influences soil wettability (Dekker and Ritsema, 1994; Ellerbrook et al., 2005), sorption of phosphates (Giesler et al., 2005), formation of metal-organic complexes (Gerke and Herman, 1992), cycling of nutrients (Qualls and Haines, 1991), acid-base chemistry of soils and water (David and Vance, 1991), leaching (Stevenson, 1994), microbial activity (Williams et al., 2004) weathering and soil formation Raulund-Rasmussen et al. (1998).
These roles of SOM become more pronounced when it is in a dissolved state. The mobility of Dissolved Organic Matter (DOM) in soils is greatly influenced by sorption to mineral sites (McDowell and Likens, 1988). Movement of Dissolved Organic Carbon (DOC) in mineral horizons of soils results in the transfer SOM, metals, nutrients and pollutants through the soil to the sub-surface and ground waters (Liu and Gary, 1993) and consequent water pollution.
Certain edaphic properties are known to relate in characteristic ways with DOC. Concentrations of DOC in soil solution typically decrease as soil depth increases (Johnson et al., 2000). It has been reported that a reduction in concentration of DOC is accompanied by a change in its composition, with preferential decrease in hydrophobic DOM. It was also observed that hydrophilic compounds sorb minimally on soil mineral surfaces than hydrophobic compounds (Kaiser et al., 1996). Sorption of DOM in soil mineral surfaces are also influenced by Soil Organic Carbon (SOC) concentration mineralogy of clay fraction, Fe and Al oxides and hydroxides (Donald et al., 1993). Sorption of SOM to iron oxides (Gu et al., 1994) and bulk soil (Kennedy et al., 1996) increased with decreasing pH as a result of increasing positive charges on the hydroxides. Given the role of SOC and SOM in low-input tropical agriculture and soil ecosystem stability amidst an increasing population, it becomes necessary to (1) determine the SOC sorption characteristics of a semi-arid environment and (2) relate its sorption properties to selected soil properties. These investigations become necessary due to climate change resulting from anthropogenic activities in his bid to satisfy his basic needs.
MATERIALS AND METHODS
Study Area
This study was conducted before the on-set rainy season in 2006 at Zamfara Grazing Reserve in Zamfara State, Nigeria in the sub-Saharan Africa. The Zamfara Grazing Reserve (6° 201 N; 7° 201 E) is covering a total area of 2,355 km2 and is located in Zamfara State, Northwestern Nigeria. It is characterized by a semi-arid climate with a uni-modal rainy season. Total annual rainfall of the area averages about 700 mm with temperature ranging from a minimum of 17°C in the months of December/January to a maximum of 40°C in April/May. Zamfara Grazing Reserve is mainly covered by Alfisols from crystalline rocks of the Precambarian basement complex (Ahn, 1979). Soils of the study area are well-drained possibly due to sandiness.. The dominant vegetation of the study area is Sudan Savannah, comprising Tamarindus indica, Adansonia digitata, Phlostigma reticulatum, Combretum nigricans, Brachiaria distichophylla and Cassia tora.
Field Studies
The first soil profile pit was located at the Grazing Reserve, the second was at the boundary with adjoining lands and the third in an adjacent grassland. These profile pits were aligned using a transect at equal distances of 500 m.
Soils are well-drained, coarse-loamy, mixed mesic Typic Haplustult (Anonymous, 2003). Three soil profile pits were dug and sampled. Soil samples were collected based on horizon differentiation. Five major horizons were identified, namely A, E, Bt1, Bt2 and Bt3. Roller tape was used to measure thickness of soil horizon. The soils were air-dried, crushed and sieved to pass through 2 mm sieve.
Laboratory Studies
Particle size distribution was obtained by the hydrometer method (Gee and Or, 2002). Soil pH was determined electrometrically at a soil to solution of 1:1 as described by Hendershot et al. (1993).
Total carbon, nitrogen and hydrogen concentrations of soils and isolated SOM were determined on ground subsamples with a CHN elemental analyzer (Thermo Quest, CE Instruments, Rodeno, Italy). Organic matter bound iron and aluminium were analyzed by sodium pyrophosphate extraction (Loeppert and Inskeep, 1996). Iron and aluminium in crystalline and amorphous oxides and hydroxides were determined by dithionite-citrate-bicarbonate extraction method (Mehra and Jackson, 1960). Aluminium and iron concentrations in the extracts were determined by graphite furnace and flame atomic absorption spectrophotometry, respectively (Perkin Elmer Corp, Norwalk, CT). Pyrophosphate extractable carbon was obtained by the UV persulfate oxidation method with a Total Organic Carbon (TOC) analyzer (Dohrman Phoenix 8000, Rosemount Analytical, Santa Clara, CA). Clay minerals were determined by X-ray diffractometry with a SIEMENS D500 diffractometer, using Ni-filtered CuK-a radiation after various pre-treatment. Semi quantitative estimation of minerals in fine-earth fraction diffractograms were done using the computer package DIFFRAC AT V3.3 SIEMENS 1993. Natural SOM was extracted from O-horizons of the profile pits. Then, field-moist soil samples were sieved through 2 mm sieve and stored at 3°C. The extraction was carried out at room temperature before sorption experiments in which 0.1 M NaOH was added to an aliquot of soil at a ratio of 1 g of soil to 20 mL NaOH for O-horizon soil; 1 g of soil to 10 mL NaOH for lower horizons and shaken for 30 min. The suspension was allowed to stand for 18 hours before they were centrifuged. The suspension was acidified using 1 μM HCl, centrifuged and the supernatant filtered through a 0.45 μm membrane filter.
The SOM was fractionated according to the procedure of Vance and David (1991) into hydrophilic and hydrophobic fractions. The extracted soluble SOM, acidified to pH 2, was pumped through a column packed with acid-washed Amberlite XAD-8 resin (Rohm and Haas Co., Philadelphia, PA). The carbon load on the resin was equal to 1.0 mg C cm-3. The soluble SOM adsorbed at pH 2 was considered hydrophobic, whereas the SOM passing XAD-8 column was regarded as hydrophilic. The hydrophilic and hydrophobic fractions were pumped through a column of strongly acidic cation exchange resin (AG-MP 50, BioRad Laboratories, Richmond, CA), to remove cations other than hydrogen before the adsorption experiment. After this treatment, fractions were kept at 4°C in readiness for sorption experiment.
Sorption
The sorption experiments were conducted at pH 3, 4 and 5. Fifty milliliters of SOM solution were added to 5 g of soil. The suspension pH was adjusted to the appropriate pH by drop-wise addition of 0.1 M NaOH with stirring during a period of 0.5 to 2.0 h. The suspension was shaken for 24 h, centrifuged and filtered through a 0.45 mm membrane filter. The DOC concentrations were obtained using TOC analyzer.
A sub-sample of the equilibrium solution was fractionated into hydrophobic and hydrophilic fractions by columns filled with XAD-8 resins as earlier described and DOC concentrations in each fraction was measured. Also, subsamples of equilibrium solutions were used to determine iron and aluminium, using flame atomic absorption spectrometry and graphite furnace, respectively.
The initial mass isotherm approach was used to describe the SOC sorption (Novdin et al., 1986). The amount of SOC adsorbed or released per unit mass of soil (R) was plotted as a function of the initial DOC concentration (X), expressed in mmol C per kg soil:
Where:
| m | = | Slope of the linear regression line, interpreted as a partition coefficient |
| b | = | Amount of substance released from soil when a solution with zero sorbate solution concentration is added |
The R was computed as the difference between the initial amount of SOC added and the amount present in the equilibrium solution.
RESULTS
Particle Size Distribution
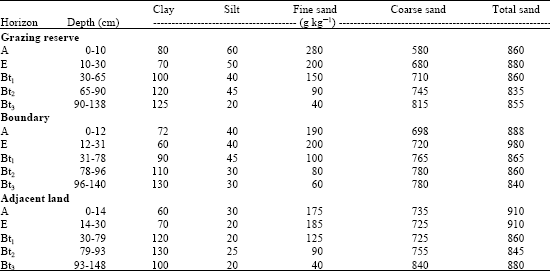
Soils were deep and dominated by sand-sized particle (Table 1). Although depth characteristic was similar, thickness of A-horizon increased away from the Grazing Reserve. Argillation was observed but occurred in deeper horizons of studied soils. Generally, silt and fine sand content decreased with depth while coarseness of sand-sized particles increased with depth.
Soil Chemical Properties
Soil chemical properties determined in the study are shown in Table 2. Soil pH decreased towards the adjacent soils of the Grazing Reserve and the same trend was observed in results of TOC, SOM and TN. Values of organic components (TOC, SOM and TN) were highest at a depth range of 30-79 cm.
| Table 1: | Particle size distribution of soils used for study |
 | |
| Table 2: | Chemical properties of soil used for the study |
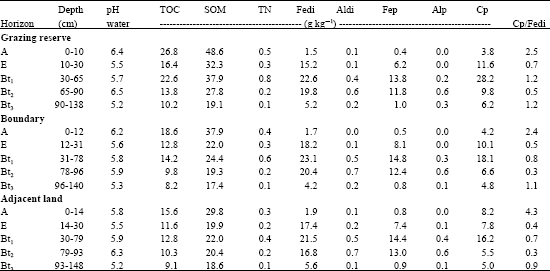 | |
| TOC = Total Organic Carbon, SOM = Soil Organic Matter, TN = Total Nitrogen, Fedi = Dithionite-citrate-extractable Iron, Aldi = Dithionite-citrate Extractable Aluminium, Fep = Pyrophosphate-extractable iron, Alp = Pyrophosphate-extractable aluminium, Cp = Pyrophosphate extractable carbon | |
In all horizons, dithionite-extractable Fe (Fedi) was significantly greater than dithionite-extractable Al. Earlier, Loeppert and Inskeep (1996), noted that dithionite is a strong reducing agent, which extracts free iron oxides, with small contributions from organically bound, exchangeable and water-soluble Fe. Also, values of pyrophosphate extractable Fe were higher than those of pyrophosphate extractable Al while pyrophosphate extractable C (Cp) ranged from 3.8 to 28.2 g kg-1. The Cp/Fedi ratios were highest at surface horizons.
Organic Carbon in Soil Horizons
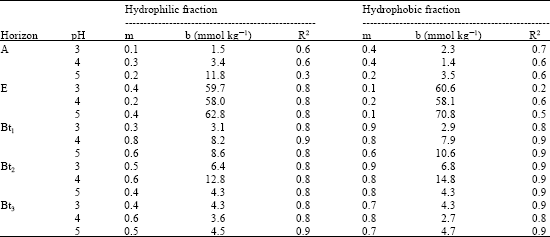
The coefficients of determination (R2) of the linear regression between the initial SOC addition and the amount of total OC released or retained ranged from 0.2 to 0.9 (Table 3, 4). Lower R2 values were observed in E-horizons of soils.
Organic C sorption was dependent on soil reaction, having maximum SOC adsorption observed in pH 4. This trend was consistent with results in both hydrophilic and hydrophobic fractions of soil OC extracted from A- and B- horizons of soils. Generally, the hydrophobic fraction showed greater sorption than the hydrophilic at all pH values in the studied soils.
| Table 3: | Initial mass isotherm parameters for the adsorption of SOC fractions extracted from the horizons (mean values) |
 | |
| Table 4: | Initial mass isotherm parameters for the SOC fractions extracted from subsoil horizons (mean values) |
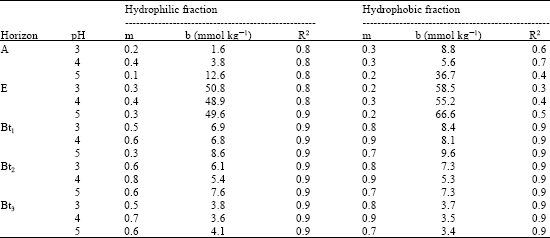 | |
The amount of substance released from soil when a solution with zero sorbate concentration was added (b) ranged from 1.4 to 70.8 mmol C kg-1 (Table 3). The released SOC (b) was lower for pH 4 suspension in all the horizons. Also the b-value increased with decrease and increase in soil pH as indicated in Table 3, 4.
The partition coefficients (m) which is a measure of the affinity of the sorbent for the substance (SOC) being adsorbed in soils ranged from 0.1 to 0.8 and 0.3 to 0.9 for hydrophilic and hydrophobic SOC fractions, respectively. The m-values ranging from 0.15 to 0.78 in Canadian soils (Moore et al., 1992) and 0.01 to 0.86 for temperate soils (Kaiser et al., 1996) have been reported. The m-values of the initial mass isotherm relationships showed that hydrophobic fraction was more strongly adsorbed than the hydrophilic fractions in all soil horizons of the studied pedons except in E-horizons. Again, SOC extracted in B-horizons had higher partition coefficients (m-values) than that obtained from A-horizon (Table 3, 4). In addition, m-values were larger when experiments were performed at pH 4 for both hydrophilic and hydrophobic SOC fraction in all B-horizons of soils as shown in Table 3, 4.
The nature of soil depth and particle size distribution in the studied soils indicate similarity in parent material and pedogenic processes (Table 1). However, land use history may have affected the variability in thickness of A-horizon among the pedons, with Grazing Reserve having the least, possibly due to weight of livestock. Earlier, Wang et al. (2001) noted that land use influences soil properties, including depth of especially A-horizon. As soils are deep, it implies that concentrations and fluxes of dissolved organic carbon in soil solution are affected, typically decreasing with increasing soil depth (Johnson et al., 2000), although this depends on the nature of the dissolved organic matter (Kaiser and Zech, 1997). Higher m-values were recorded in deeper horizons and this could be attributed to high clay content and the associated surface area. This is consistent with the observations of Kaiser et al. (1996), that sorption of dissolved organic materials in soils is affected by surface area, thus E-horizon have least m-values in the studied soils.
The E-horizon in the study area exhibited the lowest affinity for SOC sorption and this could be due to eluviation of amorphous iron and aluminium oxides and hydroxides (Table 2). Jardine et al. (1989), showed that removal of iron oxides and hydroxides using dithionite treatment of soils resulted in a significant decrease in dissolved organic carbon sorption in B-horizons of an Ultisol.
High SOM in surface horizons reduced sorption of SOC in line with the findings of Kaiser et al. (1996) and Kaiser and Zech (1997). The SOM coatings are capable of masking and blocking active sorption sites in soils, resulting in declining sorption of SOC. It implies that high concentration of indigenous SOC present in solid state reduces absorbability. Low Cp/Fedi ratio favour SOC sorption, indicating that in the studied soils, minimal sorption values obtained at the surface horizons were due to high Cp/Fedi ratios. High Cp/Fedi ratio is suggestive of the saturation of SOC binding sites in oxidic and hydrooxidic minerals.
The SOC sorption was strongly pH dependent with maximum sorption of both hydrophilic and hydrophobic fractions occurring at pH 4. in all the horizons. In a similar study, Vance and David (1991) reported mean pka values for hydrophobic and hydrophilic acids ranging from 3.8 to 4.8 for DOM originating from a Spodosol forest floor in a northern hardwood forest. Also, David and Zech (1990) observed decreasing dissolved organic carbon adsorption in the B-horizon of acid soils with decreasing pH. Decrease in sorption at lower pH of 3 could be due to soluble complex formation with iron and aluminium oxides which are more soluble at lower pH while reduced sorption at pH 5 is attributable to decreasing positive change of oxide surfaces.
Results of the study show that sorption of SOC was least in the topmost and eluvial horizons but increased in the argillic and illuvial horizons. Sorptivity was possibly influenced by the electrostatic attraction between deprotonated carboxyl groups and positively charged iron and aluminium oxyhydroxidic surfaces. The SOC adsorption decreased at lower and higher pH values, corresponding to lower charge density of the carboxyl groups and reduction in negative charge of oxyhydroxidic minerals, respectively.