Review Article
Nanotechnology: Scope and Application in Plant Disease Management
Department of Plant Protection, Aligarh Muslim University, Aligarh 202002, India
Tanveer Fatima Rizvi
Department of Plant Protection, Aligarh Muslim University, Aligarh 202002, India










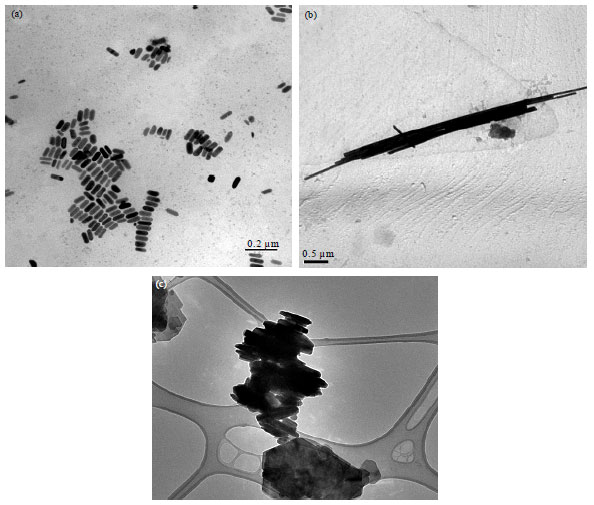


Mohamed A. Elwakil Reply
The article is not appreciated. Why?
Because nano-particles end up on rivers and lake beds. It is expected that these particles change the composition of water life including the reproduction and life cycle of water creatures. On the other hand, treating plants with nano-particles (fertilizers and pesticides) will be integrated in the food chain and finally accumulated in the human body. No settled studies on the diverse effect of nano-particles in agriculture are available. So far, this article is not appreciated as the use of nano-particles in agriculture is still a questionable issue at the time being.
M. R. Khan
I appreciate your viewpoint. The concern you have raised about the environmental issues is important and worth considering, hence has already been discussed in the paper on page 224-225 under �Biosafety, the Environmental risks and Conclusion�. Nanotechnology is in its initial stage, plant protectionists do need to explore the use of new/novel materials such as nano forms of matter to deal with the plant diseases and pesticide contamination, not with an approach to generate a new problem to control a problem. Nanopesticides, if found suitable may drastically reduce the amount of the pesticide required to control a plant disease. With this approach, efforts to explore possibility of using nanomaterials in the plant disease management should be appreciated. It is not wise that use of a novel material is not explored due to apprehension of its adverse effects suspected. Science works on the principle of continuous efforts of exploration, invention and improvement, a few biodegradable nanomaterials are on target, which may replace metallic nanomaterials, if problems are visualized.
David Hyde
Nano beads in cosmetics, they already cause a consequence.
Prof. Bal Ram Singh Reply
Going through the article, I enjoyed reckoning the future potential of nanoscience in agriculture and its allied disciplines, the plant pathology. Nanoscience is currently at a stage where it may not be categorized as a merititful or disadvantageous technology. In view of pace of advancements in this discipline, the nanoscience is likely to play a major role in plant disease management as well as in animal nutrition and aquaculture. Prof. B.R.Singh
Dr. N.V. Singh Reply
The article is good and gives upto date information of the utility of nano forms in the disease control. The articles provides a platform to judge the merits and demerits of nano science in agriculture. The articles has highlighted a number of ill-effects of nanoparticles which must be considered while devising future strategies for nano application in plant sciences.
Ganpat K. Kavimadam Reply
Being optimistic, the Nanotechnology might have a beneficial impact on agriculture production system through a variety of means, plant disease control may be one of them. However, in view of limited information on these aspects, and to minimize the chances of unpredicted future risks, international standards for nanotechnology are required to be setup. Further, differences in the approach of various nations towards nanotechnologies need to be resolved. However, nanotechnology have shown indications of socioeconomic prospects, particularly for the developing and agriculture based countries, if adequate infrastructure to verify/ deal with the toxicological aspects is developed. Dr. G.K. Kavimadam
Dr. Sudha Rani Reply
Nano Technology may make a breakthrough in crop production. Papers and reviews in this area may help in exploring its strength of novelty and utility in pest control. Even to establish unsuitability of nano materials in disease control, such contributions are equally important. The contents of the paper deserve recognition & appreciation. Sudha
Dr. Mohd. Anis Ansari Reply
We do not know the future course of advancement. It is hoped that the toxicity aspect of nano materials will be solved, and this science will be used to protect crops and to improve their productivity.
Prof. D.S. Prasad Reply
Yes, there is substantial apprehension of contamination of soil and water by nano-materials. However, remote possibvility exists of occurrence of such situation to an extent that becomes of major concern. I am in favor of test and trial of nano-particles in crop production system. Prof. Prasad
Prof. Ram B.Singh Reply
Excellent paper. Aoutor deserve appreciation for gathering huge information and presenting in an organised manner. This review article is probably first in a way that makes aware a reader about history, methods of production of nanoparticles and possibilities on application of these tiny particles to control plant diseases. The article may form a basis for future advancements in plant disease management including the IPM.