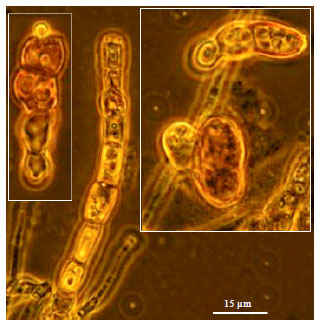
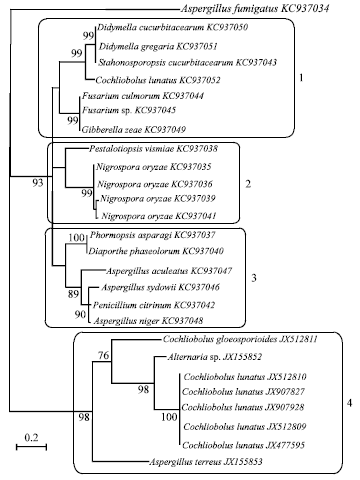
Research Article
Diversity of Ascomycetes at the Potato Interface: New Devastating Fungal Pathogens Posing Threat to Potato Farming
Department of Biochemistry, University of Yaounde I, BP812, Yaounde, Cameroon
Sayanika D. Waikhom
Institute of Bioresources and Sustainable Development, Takyelpat, 795001, Imphal, Manipur, India
Wakambam M. Singh
Institute of Bioresources and Sustainable Development, Takyelpat, 795001, Imphal, Manipur, India
Narayan C. Talukdar
Institute of Bioresources and Sustainable Development, Takyelpat, 795001, Imphal, Manipur, India
Pranab Roy
Department of Biotechnology, Haldia Institute of Technology, 721657, Haldia, West Bengal, India