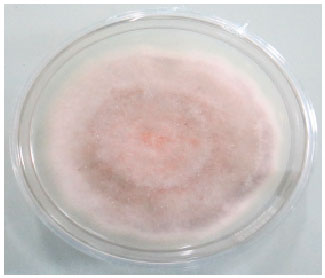
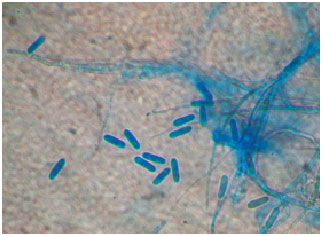
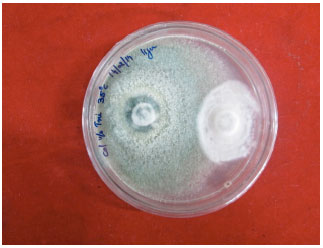
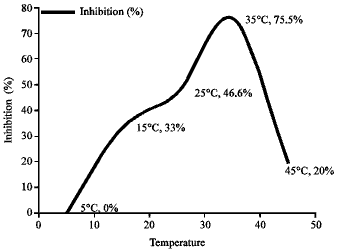
Research Article
Control of Anthracnose Disease in Swietenia macrophylla using Trichoderma virideas Biocontrol Agent
Institute of Forest Genetics and Tree Breeding, Coimbatore, 641002, India
A. Karthikeyan
Institute of Forest Genetics and Tree Breeding, Coimbatore, 641002, India
N.B. Siva Priya
Institute of Forest Genetics and Tree Breeding, Coimbatore, 641002, India
Sadia Siddiqui
Institute of Forest Genetics and Tree Breeding, Coimbatore, 641002, India