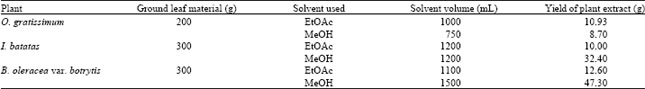
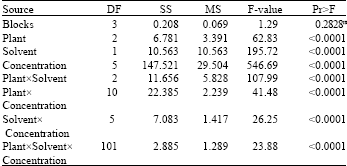
Research Article
Effects of Selected Plants’ Extracts on in vitro Growth of Ralstonia slanacearum (Smith), the Causal Agent of Bacterial Wilt of Irish Potatoes
National Museums of Kenya, P.O. Box 40658-00100, Nairobi, Kenya
S. O. Wagai
Maseno University, P.O. Box 333-40105, Maseno, Kenya
L. Manguro
Maseno University, P.O. Box 333-40105, Maseno, Kenya
B. M. Gichimu
Coffee Research Foundation, P.O. Box 4-00232, Ruiru, Kenya