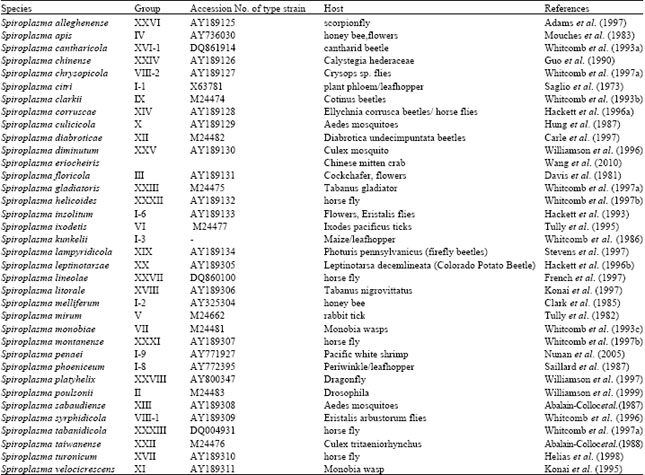
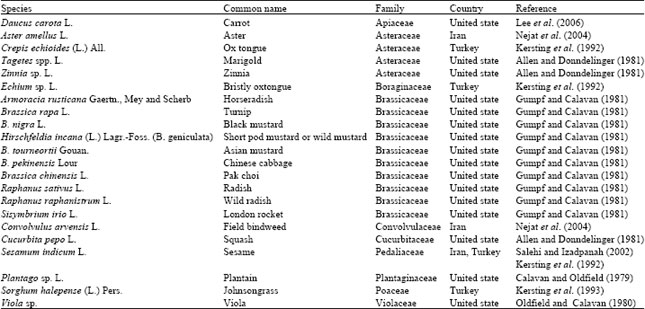
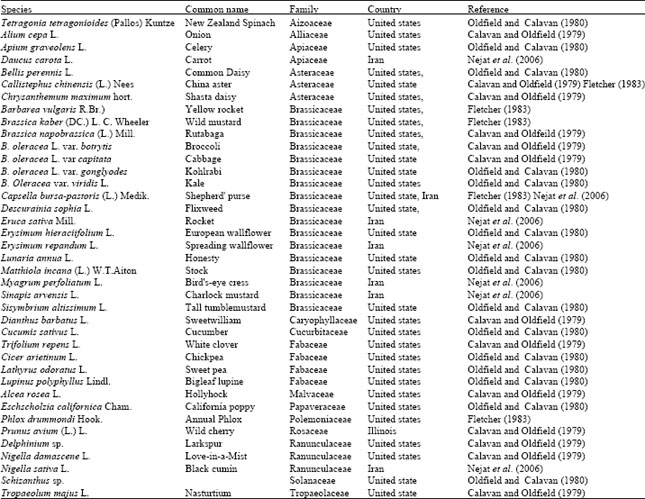
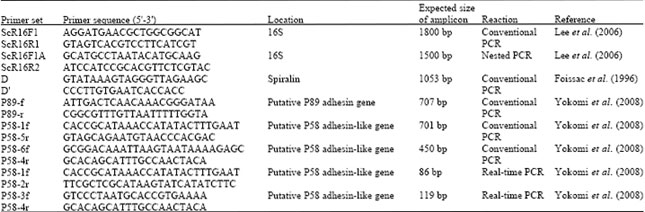
Review Article
Spiroplasma citri: A Wide Host Range Phytopathogen
Institute of Tropical Agriculture, Universiti Putra Malaysia, 43400, Malaysia
G. Vadamalai
Department of Plant Protection, Faculty of Agriculture, Universiti Putra Malaysia, 43400, Malaysia
M. Dickinson
School of Biosciences, University of Nottingham, Sutton Bonington Campus, Loughborough, LE12 5RD, UK