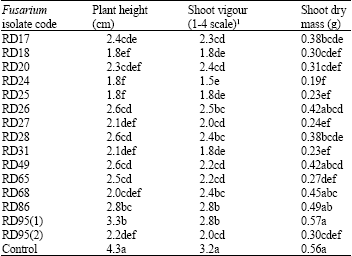
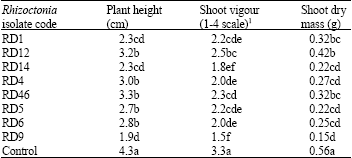
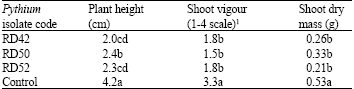
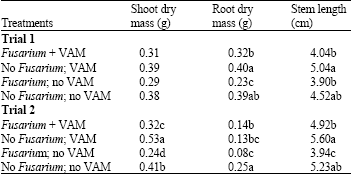
Research Article
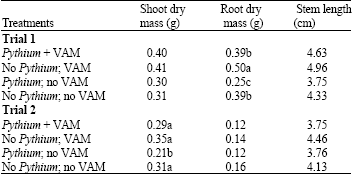
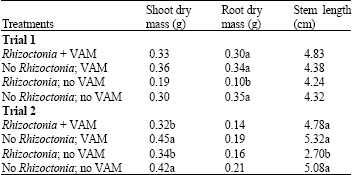
Causal Agents of Root Rot and the Effect of Vesicular-Arbuscular Mycorrhizal Fungi in Seedlings of Rhodiola rosea in Alberta, Canada
Crop Diversification Centre North, Alberta Agriculture and Rural Development, Edmonton, AB, T5Y 6H3, Canada
H.U. Ahmed
Crop Diversification Centre North, Alberta Agriculture and Rural Development, Edmonton, AB, T5Y 6H3, Canada
K. Ampong-Nyarko
Crop Diversification Centre North, Alberta Agriculture and Rural Development, Edmonton, AB, T5Y 6H3, Canada
S.E. Strelkov
Department of Agricultural, Food and Nutritional Science, University of Alberta, Edmonton, AB, T6G 2P5, Canada
R.J. Howard
Crop Diversification Centre South, Alberta Agriculture and Rural Development, Brooks, AB, T1R 1E6, Canada
G.D. Turnbull
Crop Diversification Centre North, Alberta Agriculture and Rural Development, Edmonton, AB, T5Y 6H3, Canada