ABSTRACT
The capability of Urocystis agropyri as a bioagent to control ryegrass in wheat fields was examined. Non-significant difference between the wheat production sown in soil inoculated and non-inoculated with the isolate of U. agropyri, but highly differences were recorded when wheat and ryegrass sown together in soil inoculated and non-inoculated with this isolate. The results indicated that ryegrass had a significant negative effect on the productivity and yield components of wheat. The productivities of wheat were 2402 and 2362 kg ha-1 for only wheat sown in soil non-inoculated and inoculated with U. agropyri, while they were 1184 and 690 kg ha-1 for wheat and ryegrass sown in soil inoculated and non-inoculated with the isolate of U. agropyri, respectively. This confirms that ryegrass had a significant negative effect for wheat productivity and the isolate of U. agropyri can be used as a biological agent to control this weed in order to increase the productivity of wheat.
PDF Abstract XML References
How to cite this article
DOI: 10.3923/ppj.2007.260.265
URL: https://scialert.net/abstract/?doi=ppj.2007.260.265
INTRODUCTION
Weeds are usually recognized as unwelcome plants, because they pose a recurrent and ubiquitous threat to agricultural productivity. They may represent shelters for disease victors, as well as compete with the economic crops for moisture, nutrient and light. Moreover, weeds significantly affect human health through allergenic reactions, skin irritations, mechanical injury, or internal poisoning (Ziska, 2001). Weed losses and weed control have an estimated annual economic cost of more than $ 15 billion in the USA alone (Bridges, 1994). Several annual and perennial grasses are among the most problematic weeds in various crops in several countries (Holm et al., 1997; Boari and Vurro, 2004). Annual ryegrass, Lolium multiflorum Lam., is widely spread in wheat field located in Al-Qassim region, Central Saudi Arabia and can inhibit the growth of wheat and delay grain production (Shaukat and Mouafaq, 1983; Kawas and Moshileh, 2003). This weed is difficult to control, because its tolerance to available chemical herbicides or because its growth habits enable it to escape from chemical and mechanical control practices (Chandramohan and Charudattan, 2001). The discovery and development of host specific fungal plant pathogens as bioherbicides may provide a nonchemical option to manage weedy grasses (Charudattan, 1991; Templeton, 1990). Urocystis agropyri (G. Prenss) (Syn. Urocystis tritici Korn) caused flag smut disease in wheat, barley and several grasses including bent grasses (Agrostis sp.), meadow grasses (Poa pratenis), tall oat grass (Arrhenatherum elatives), fescues grass (Festuca arundinacea), agropyron grass (Agropyron repens) and ryegrass (Lolium sp.) (Nus and Hodges, 1990; Mathur and Barry, 1993).
unfortunately, there is no widely bioherbicide commercially available to control ryegrass, although some fungal pathogens have been reported as potential candidates for others (Massion and Lindow, 1986; Figliola et al., 1988; Chiang et al., 1989; Zhang and Watson, 1997). In the selection of agents for the biological control of weeds, the selected organisms should be sufficiently specific to the weed host and are nonpathogenic to plants of economic importance (Smith, 1986; Doreete and Jurgen, 2005). The present study aimed to (i) identify and characterize plant pathogen U. agropyri isolate collected from ryegrass growing in wheat fields located in Al-Qassim region, (ii) evaluate the bioherbicidal efficacy of U. agropyri isolate in trials under greenhouse and field conditions and (iii) determine the safety of U. agropyri isolate for selected, economically important non-target plant species.
MATERIALS AND METHODS
Inoculums Production: Mature ryegrass plants infected with flag smut disease were collected at the end of growing season 2003-2004 from Al-Qassim wheat fields located at Central Saudi Arabia and stored in the refrigerator until used as a inoculum source. Fungal inoculums was prepared by dipping infected ryegrass plants for 2 h in 1000 mL Erlenmeyer flasks containing sterilized distilled water and then passed through a single layer of cheesecloth. Spore balls concentration was adjusted with the aid of hemocytometer to 1.9x108 spore balls mL-1.
Pathogenicity test
Effect of inoculums concentration of U. agropyri on ryegrass: Plastic pots (18 cm) were filled with autoclaved soil, sown with ten seeds of ryegrass per each and then inoculated with U. agropyri at the rates of 1, 5, 10, 25, 50 and 100 mL per pot. At the same time another pots were non-inoculated with spores and used as a control. Experiment was replicated four times.
Effect of U. agropyri on ryegrass under greenhouse and field: In greenhouse, pots were filled with autoclaved soil, sown with ten seeds of ryegrass and inoculated with 50 mL of U. agropyri per pot. Under the field conditions, plot (2x3 m2) was sown with ryegrass, each row received 200 grain and inoculated with 50 mL m-1 of U. agropyri. Experiment was replicated four times. The flag smut disease and the percentage of infection were determined at heading stage.
Greenhouse experiments
Effect of U. agropyri in wheat cultivars and ryegrass: Twenty wheat cultivars and one ryegrass were used in the greenhouse study (Table 1). Pots were filled with autoclaved soil, received ten grains from the cultivars per pot and then inoculated with U. agropyri. Control treatment was non-inoculated with U. agropyri. Experiment was replicated four times.
Host range determination: Host range determination were done for U. agropyri, included several grasses that have problematic weed in wheat fields. Five plant species
namely, wheat (Yecora rojo cultivar), barley (wild type), avena (Avena sterilis), setaria (Setaria pumila) and ryegrass (Lolium multiflorum) were used. Pots were filled with autoclaved soil, each pot received 5 grains from each plant species and inoculated with U. agropyri. Non-inoculated grains were used as control. Experiment was replicated four times. The flag smut disease was determined at the heading stage.
Field experiments
Effect of U. agropyri in wheat and ryegrass cultivars: Twenty wheat cultivars and one ryegrass collected from local and international source were tested. Field experiment was conducted at the Agricultural Experimental Station, Al-Qassim University, Central Saudi Arabia during the growing season 2004-2005. The experimental layout was Complete Randomized Block Design (CRBD) with four replicates for each treatment. The treatments included soil inoculated and the other soil non-inoculated with U. agropyri. Plots (1x4.2 m2, twenty one rows) were sown with either wheat cultivars or ryegrass (100 grain per row). Fungal inoculums was added to soil at the rate of 50 mL per row. The flag smut disease was determined at heading stage. Experiment was replicated four times.
Effect of ryegrass and U. agropyri on the production of wheat: The experimental layout was a (CRBD) with four replicates for each treatment. The experiment included four treatments as following: 1) wheat sown in soil inoculated with U. agropyri; 2) wheat sown in soil non-inoculated with U. agropyri; 3) wheat and ryegrass sown in soil inoculated with U. agropyri and 4) wheat and ryegrass sown in soil non-inoculated with U. agropyri.
| Table 1: | Effect of Urocystis agropyri on the wheat and ryegrass cultivars cultivated under greenhouse and field conditions |
 | |
The rate of inoculum was 50 mL m-1. Plot area was 2x3 m2. The most commonly cultivated cultivar in Saudi Arabia, Yecora rojo and ryegrass cultivar (Lolium multiflorum) were used at the rate of 100 grain m-1. Near maturity, the number of wheat plants, number of spike in one square meter and the dry weight of 10 wheat plants were determined in each plot. At maturity stage, the central five rows were hand harvested and yielded.
RESULTS AND DISCUSSION
Pathogenicity and characterization of U. agropyri: Data in Table 1, show that the isolate of U. agropyri was highly pathogenic to ryegrass under either greenhouse or field conditions, where the percentages of infection were 35.8 and 19.2%, respectively. On the contrary, this isolate did not infect any cultivars of the investigated wheat under greenhouse and field conditions.
Ryegrass plants collected from the field and naturally infected with U. agropyri were dried and did not form either spike or grains (Fig. 1A). The spore mass were dusty, dark brown to black composed of spore balls only, which oblong to sub-spherical, 18-35 Um diam, each composed of 1-5 (usually 1-3) spores almost completely surrounded by hyaline to yellowish thin-walled, oblong to sub-spherical sterile cells 3-12 Um across (mean 6.6) Um diam. Spore subylobose or slightly flattened 11-20 (mean 15.5) Um (Fig. 1B). The effect of inoculum concentrations of U. agropyri to induce flag smut disease in ryegrass was shown in Table 2. It was found that there were significant differences between the control and soil infected of either 1 or 5 mL spore balls of U. agropyri as well as the disease did not appear. The disease appeared when pots received either 10, 25, 50 or 100 mL spore balls and severity of the disease was concentration dependent. The corresponding percentages of infection were 14.6, 24.5, 37.4 and 56.4%, respectively.
The pathogenicity test showed that flag smut disease of ryegrass appeared when the soil inoculated with spore balls of U. agropyri under either greenhouse or field conditions, where the percentages of infection were 36.5 and 19.9%, respectively (Table 3). The symptoms of flag smut in ryegrass were reported during either the growth of the plant (Fig. 2A) or at the end of maturity stage (Fig. 2B). Symptoms appeared as small white or grayish, blister-like streaks depend on close examination of the primary leaf at the seedling stage plants inoculated with U. agropyri. Flag smut is most conspicuous in the late spring. During stem elongation and heading stage of growth long grayish-black streaks of sub-epidermal smut spori develop between the veins of leaf blades and sheaths. At the plant maturity, the spori enlarge and bread through the epidermis exposing black spore masses. Infected plants are usually stunted with distorted and twisted leaves and have more tillers than healthy plants. When the disease is very severe, ears did not develop or prevented them from emerging by the twisting of the leaf whorl, (Fig. 2A and B). These symptoms were observed also by many investigators (Singh, 1982; Mordue and Ainsworth, 1984; Mathur and Barry, 1993).
 | |
| Fig. 1: | (A) Natural infection of flag smut in ryegrass in the field, (B) the spore balls of Urocystis agropyri each composed of 1-5 (usually 1- 3) spores |
| Table 2: | Effect of different inoculum concentrations of U. agropyri to induce flag smut disease in ryegrass |
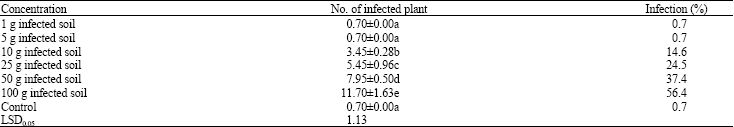 | |
| Data are expressed as mean, Means within the same column and followed by the same coefficient are not significant different from each other (p≤0.05) | |
| Table 3: | Pathogenicity test of U. agropyri on ryegrass under greenhouse and field conditions |
 | |
| Data are expressed as mean, Means within the same column and followed by the same coefficient are not significant different from each other (p≤0.05) | |
 | |
| Fig. 2: | (A) The artificial infection of flag smut in ryegrass during maturity, (B) at the end of maturity stage, (C) cleared that the isolate of U. agropyri infected only ryegrass (1= avena, 2= setaria, 3 = barely, 4 = wheat and 5 = ryegrass) |
Host range determination: The host range of the isolate of U. agropyri collected from Al-Qassim fields in Central Saudi Arabia was tested under greenhouse conditions in five plant species which have problematic in wheat field. The results show that, this isolate did not invasive wheat (Yecora rojo), barley (wild type), Avena sterilis and Setaria pumila but infected only ryegrass (Lolium multiflorum ) (Fig. 2C). The host range of U. agropyri caused flag smut in wheat, barley, bent grasses, meadow grasses, tall out grass, fescues grass, agropyron grass and ryegrass (Mordue and Ainsworth, 1984; Nus and Hodges, 1990; Rossmam et al., 1990). The present results cleared that the isolate of U. agropyri isolated from Al-Qassim wheat fields in Central Saudi Arabia, not infected the cultivars of tested wheat or the tested weeds, but infected only ryegrass (Table 1 and Fig. 2C). The physiological races of the U. agropyri have been reported from several countries included 12 from China and 2 from the USA (Sing, 1982).
| Table 4: | Effect of ryegrass and U. agropyri on the production of wheat in field |
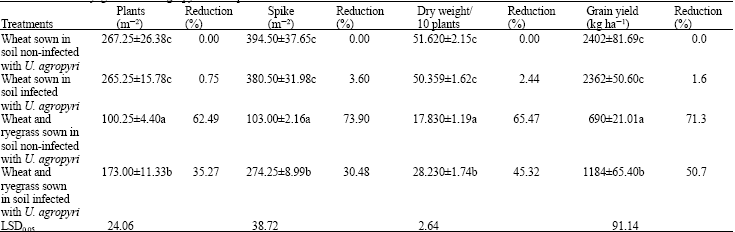 | |
| Data are expressed as mean. Means within the same column and followed by the same coefficient are not significant different from each other (p≤0.05) | |
Moreover, inoculation of U. agropyri collected from Baghdad in Iraq represents a different physiological races against some cultivars of wheat (Al-Beldawi et al., 1987).
Effect of ryegrass and U. agropyri on wheat production: Table 4 shows that there were no significant effects concerning spikes m-2, dry weight per 10 plants and grain yield of wheat between the wheat sown in soil infected or non-infected with the isolated of U. agropyri but the significant differences were observed when wheat and ryegrass sown in soil infected or non-infected with the isolate of U. agropyri. The isolate of U. agropyri infected ryegrass only in wheat field. However, the reduction percentages of wheat plants were 35.27 and 62.49% when soil infected and non-infected with U. agropyri, respectively. The percentages reduction in spikes m-2, dry weight per 10 wheat plants and grain yield (kg ha-1) were 30.48, 45.321 and 50.7% in soil infected and 73.9, 65.466 and 71.3% in non-infected soil with the isolated of U. agropyri, respectively.
These results clearly indicated that the isolate of U. agropyri infected ryegrass and affects its growing which let wheat plants to give high production. The present results are in parallel with that obtained by Kawas and Moshileh (2003) who reported that ryegrass had significant negative effect on the productivity and yield components of barley.
It can be concluded that this isolate of U. agropyri can be used as a biological agent in wheat fields to control ryegrass in order to increase wheat productivity.
REFERENCES
- Chandramohan, S. and R. Charudattan, 2001. Control of seven grasses with a mixture of three fungal pathogens with restricted host range. Biol. Control, 22: 246-255.
CrossRefDirect Link








