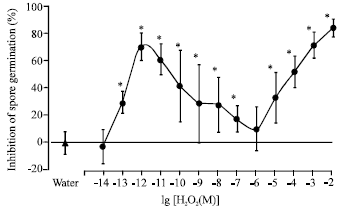
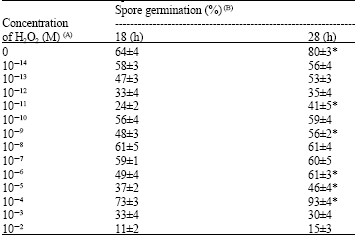
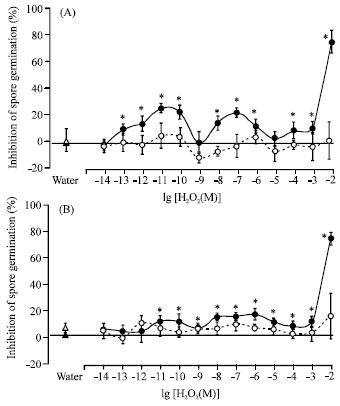
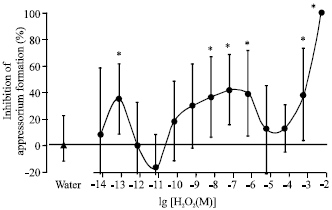
Research Article
Suppression of Early Stages of Fungus Development by Hydrogen Peroxide at Low Concentrations
Research Institute of Phytopathology, Russian Academy of Agricultural Sciences, B. Vyazemy, Moscow Region, 143050, Russia
Vera P. Lapikova
Research Institute of Phytopathology, Russian Academy of Agricultural Sciences, B. Vyazemy, Moscow Region, 143050, Russia
Tatiana D. Pasechnik
Research Institute of Phytopathology, Russian Academy of Agricultural Sciences, B. Vyazemy, Moscow Region, 143050, Russia
Vladimir V. Kuznetsov
Russian Peoples Friendship University, Faculty of Agriculture, 8 Miklukho-Maklaya str. Moscow, 117198, Russia
C. Jacyn Baker
Molecular Plant Pathology Lab., ARS USDA, Beltsville, Maryland 20705, USA