Research Article
Protease in Tomato Fruits Infected by Fusarium oxysporum
Department of Microbiology, Obafemi Awolowo University, Ile-ife, Nigeria
P.O. Olutiola
Department of Microbiology, Obafemi Awolowo University, Ile-ife, Nigeria
Tomato, one of the most important vegetables in many countries has a world-wide economic and nutritive importance. According to Esquinas-Alkazar[1], it contains an estimated value of 1.2 g of protein and an energy value of 20 Kcal/100 g of edible product. Fusarium species have been known to cause substantial global loss of the fruits especially in the tropics[2]. Infected fruits become soft and wrinkled; lesions are later produced with slightly raised white to pink mouldy growth[3].
The known group of hydrolytic enzymes capable of initiating protein degradation are the proteolytic enzymes (proteases)[4-6]. This study describes the degradation of protein tissues of Lycopersicon esculentum Miller variety of tomato fruits by protease from Fusarium oxysporum during infection process. Properties of the enzyme were also studied.
Organism and culture condition: The isolate of Fusarium oxysporum used was part of a culture collection of Prof. P.O. Olutiola, Department of Microbiology, Obafemi Awolowo University, Ile-Ife, Nigeria. The organism was routinely grown and maintained on 1% malt yeast extract-glucose agar slants in test tubes. Seventy-two-hour old cultures served as inoculum.
Inoculation of tomato fruits: Ripe tomato (Lycopersicon esculentum Miller) fruits were obtained from the Obafemi Awolowo University Teaching and Research Farm. They were surface sterilized in 1% sodium hypochlorite for 10 min and thoroughly rinsed with sterile distilled water. Using a cork borer, a tissue disc was removed from each tomato fruit and replaced with tissue discs of the inoculum. The holes were sealed with paraffin wax. Tomato fruits inoculated with sterile malt yeast extract agar served as control. Procedures were carried out in a sterile inoculating chamber. Experimental and control fruits were placed separately in sterile petri dishes on glass plates with bell jars inverted over them at 26°C. The rims of the bell jars were sealed with grease to ensure air tight contact with the glass plates. The fruits were observed daily for deterioration.
Extraction of enzyme: Within ten days of inoculation the tomato fruits had completely collapsed. The fruits were weighed, homogenized with cold (4°C) liquid extractant (2:1 w/v). The extractant consisted of 0.5 M NaC1 in 0.1M citrate phosphate buffer (pH 7). The homogenate was filtered through four layers of muslin and further clarified by filtering through filter paper (Whatman No.1). The protein content of the crude filtrate was determined as previously described by Lowry et al.[7]. Protease enzyme activity was analysed by a modified method of Kunitz[8].
Preparation of extract for fractionation: The crude enzyme was concentrated to about one sixth of its original volume in a vacuum rotary evaporator (Quick fit, Rotavapor-R, Buchi, Switzerland) at 30°C under low vacuum pressure and slow evaporation[9].
Fractionation on Sephadex G-25: The column (1.5x30 cm) of Sephadex G-25 was prepared as previously described by Olutiola and Cole[10]. It was equilibrated with 0.02 M citrate phosphate buffer pH 6.0 and equipped with a constant temperature water jacket (4°C). Ten milliliter of the concentrated enzyme was applied to the column and eluted with 0.02 M citrate phosphate buffer pH 6.0 containing 5 mM NaN3. Column fractions were collected (5 mL/tube) and optical density of the fractions were measured at 280 nm. Each fraction was analysed for protease activity.
Fractionation on Sephadex C-50: Fractions (15-17) from Sephadex G-25 which showed appreciable protease activity were pooled together. Ten milliliter of the pooled enzyme was applied to Sephadex C-50 column (2.5x40 cm) surrounded by a constant temperature water jacket (4°C), supplied by Pharmacia fine chemicals, Uppsala, Sweden.
Fractions (5 mL/tube) were eluted with 0.02 M citrate phosphate buffer pH 6.0 containing a gradient (0.02-0.03 M) NaCl. Optical density of each fraction was measured at 280 nm. Fractions were analysed for protease activity.
Fractionation on Sephadex G-100: Fractions collected from Sephadex C-50 column which showed appreciable protease activity were pooled together. Ten milliliter of the mixture was applied to Sephadex G-100 column (2.5x70 cm) which had been previously calibrated by proteins of known molecular weight as previously described by Andrews[11], Olutiola and Cole[12]. Fractions (5 mL/tube) eluted with 0.02 M citrate phosphate buffer pH 6.0 were analysed for protease activity.
Enzyme assay: Protease activity was analysed using a modified method of Kunitz[8]. The substrate, 1% (w/v) casein (Sigma) solution, was prepared in 0.02 M citrate phosphate buffer (pH 6.0). It was heat denatured at 100°C for 30 min in a water bath, cooled and filtered.
The reaction mixture was 1 mL of enzyme preparation added to 1 mL of the substrate. Incubation was at 35°C for 1 h. Reaction mixture was terminated by adding 3 mL of cold 10% (w/v) trichloroacetic acid (TCA). Controls which initially contained only 1 mL of the substrate were incubated with the experimental tubes at 35°C for 1 h. Three milliliter of cold trichloro acetic acid was added to each control tube followed by 1 mL of enzyme. After termination of reaction, experimental and control tubes were left to stand at 0°C for 1 h to aid complete precipitation of unhydrolysed protein. The supernatant was filtered and analysed for non-precipitateable protein using the Lowry et al.[7] method. One unit of protease activity was arbitrarily defined as the micro moles of non-precipitable (TCA) tyrosine released in 30 min. Specific activity was calculated as enzyme unit per mg protein.
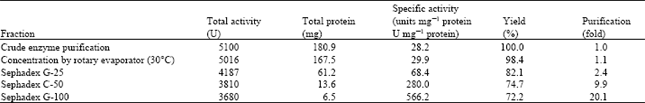
When healthy tomato fruits were inoculated with Fusarium oxysporum, they became soft and wrinkled. There was extensive degradation within ten days of inoculation. Extracts of infected fruits exhibited protease activity. Uninfected fruits did not. Gel filtration of concentrated extract on Sephadex G-25 column showed two peaks of absorption. Only the components of the first peak exhibited protease activity. Fractionation of the components of this first peak on Sephadex C-50 column gave three peaks of absorption. Only the components of the second peak exhibited protease activity. When the components of this peak was further fractionated on Sephadex G-100 column; two peaks of absorption were obtained. The molecular weight estimates of the peaks were 11200 and 63,100. Only the components of the heavier peak showed protease activity. Purification steps are represented in Table 1.
Some properties of the partially purified protease were investigated. Within a temperature range of 20 to 50°C, optimum activity was observed at 35°C. Using 0.02 M citrate phosphate buffer with pH ranges of 4.0-8.0, optimum pH was recorded at pH 6.0. The protease was stimulated by Na+, Mg++, Ca++ and K+ ions. Optimum activity was observed at 20 mM concentration of Mg++ ion whereas further stimulation in activity was observed at 30 mM concentrations for Na+, Ca++ and K++ ions. With different concentrations of casein used as substrate (ranges of 2-16 mg mL-1), optimum activity was recorded at 12 and 14 mg mL-1. The rate of enzyme reaction seemed to follow the Michalis-Menten kinetics. From the Lineweaver Burk Plot, the apparent Km for the hydrolysis of casein was approximately 4.34 mg mL-1. The protease was inhibited by ethylene diamine tetra acetic acid and mercuric chloride. There was total inhibition at 10 and 4 mM concentrations, respectively.
| Table 1: | Partial purification of protease from tomato fruits infected by Fusarium oxysporum |
 | |
Fruit rot and extensive degradation occurred during the infection of apparently healthy tomato fruits by Fusarium oxysporum. Protease activity was detected in extracts of the infected fruits. Similar observations have been previously reported on Zea mays seedlings infected by Fusarium avenaceum[13] and on soy beans infected by Aspergillus flavus var columnaris[14].
Temperature changes affected the enzyme. Optimum activity was at 35°C. A similar temperature optimum has been reported of a protease from Aspergillus aculeatus associated with the “black tongue” disease in man[15]. The enzyme showed optimum activity at pH 6.0. Hussein et al.[16] reported a similar pH optimum for the protease from the thermophilic Streptomyces thermocinereus. The enzyme was able to degrade casein with optimum activity recorded at 12 and 14 mg mL-1 concentrations. A similar report describes a protease from Escherichia coli as capable of cleaving casein into two large polypeptides (which cannot be further degraded) and smaller peptides of about 5,000 daltons[17]. Activity of the protease was stimulated by the cations Na+, Ca++, Mg++ and K+, Activity increased with increasing concentration of each cation indicating that the protease might require a metal ion activator. Takami et al.[18] reported that calcium ions stimulated the activity of a protease obtained from an alkalophilic Bacillus sp. A similar observation was reported by Tsuchiya et al.[19] on the alkaline protease obtained from an alkalophilic thermoactinomycete.
Activity of the enzyme was inhibited by ethylene diamine tetra acetic acid (EDTA) and mercuric chloride (HgCl2). Aspergillus fumigatus produces an extracellular metalloprotease which is completely inhibited by EDTA[20]. The presence of protease in tomato fruits infected by Fusarium oxysporum and its absence in uninfected tissues clearly suggests that the enzyme might play a role in the infection process.