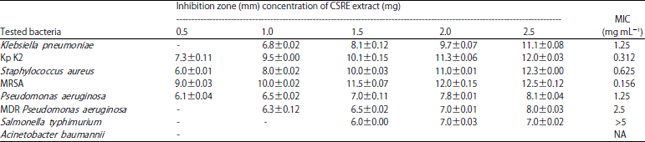
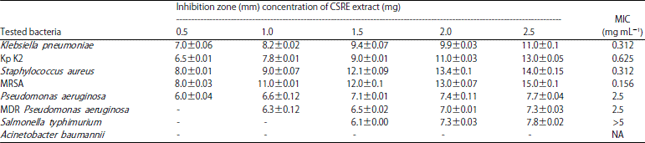
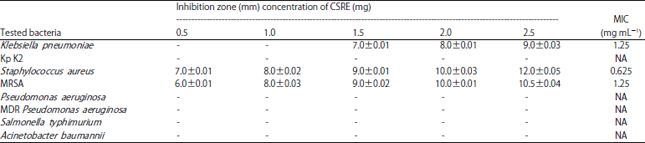
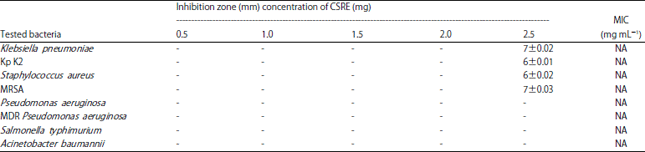
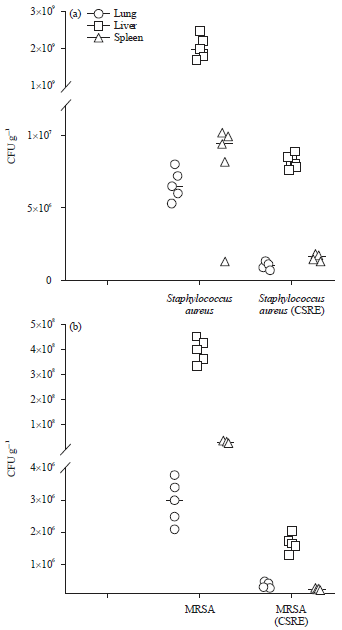
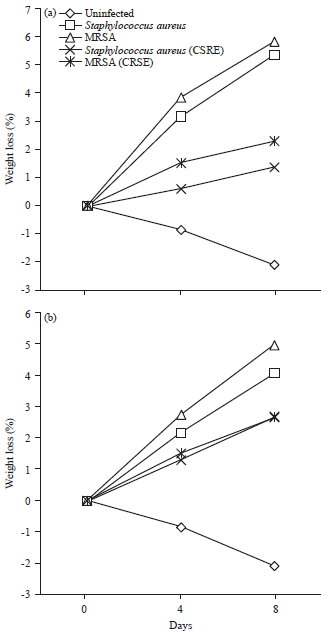
Research Article
In vitro and in vivo Antibacterial Activity of Cheilocostus speciosus Rhizome Extract on Resistant Bacteria
Department of Biology and Biotechnology, Faculty of Science, American University of Madaba, Madaba, Jordan
LiveDNA: 962.32206
Hammouh Fadwa
Department of Nutrition and Dietetics, Faculty of Health Sciences, American University of Madaba, Madaba, Jordan
Dababneh Basem
Department of Nutrition and Dietetics, Faculty of Health Sciences, American University of Madaba, Madaba, Jordan
Gammoh Omar
Department of Pharmacy, Faculty of Health Sciences, American University of Madaba, Madaba, Jordan
Al-Smadi Ahmed
Department of Medical Laboratories, Faculty of Health Sciences, American University of Madaba, Madaba, Jordan
LiveDNA: 962.12638