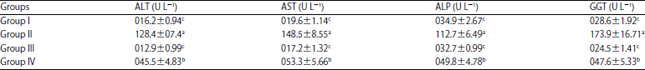
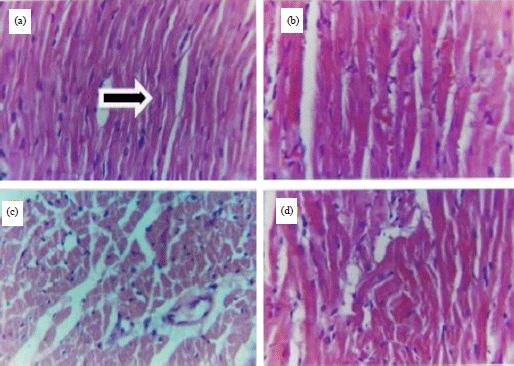
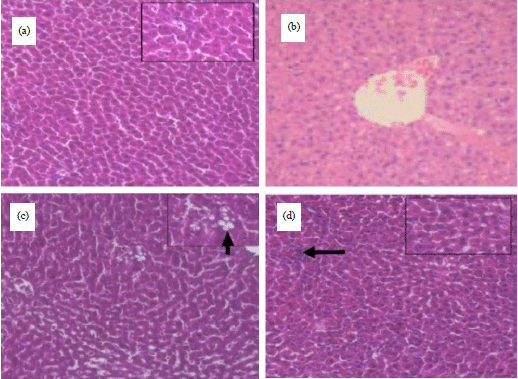
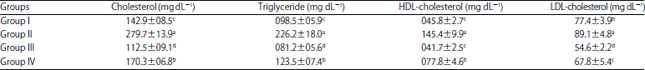Research Article
Protective Role of Juniperus phoenicea L. Leaves Extract against Gamma-irradiation-induced Oxidative Stress
Institute of Agricultural Genetic Engineering Research, Agricultural Research Center, Egypt
LiveDNA: 20.31417
Ismail A. Ismail
Institute of Agricultural Genetic Engineering Research, Agricultural Research Center, Egypt
Ehab I. El-Hallous
Institute of Agricultural Genetic Engineering Research, Agricultural Research Center, Egypt
LiveDNA: 20.32414
Walaa F. Alsanie
Department of Medical Laboratories, Faculty of Applied Medical Sciences, Taif University, Kingdom of Saudi Arabia