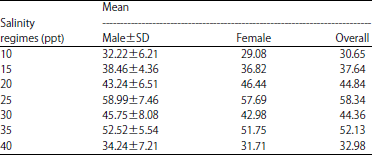
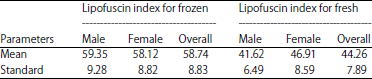Research Article
Salinity and Preservation Conditions of Lipofuscin Extracts in Blue Swimming Crab, Portunus pelagicus
Institute of Tropical Aquaculture and Fisheries, Universiti Malaysia Terengganu, 21030 Kuala Nerus, Terengganu, Malaysia
Mhd Ikhwanuddin
Institute of Tropical Aquaculture and Fisheries, Universiti Malaysia Terengganu, 21030 Kuala Nerus, Terengganu, Malaysia
LiveDNA: 60.4241