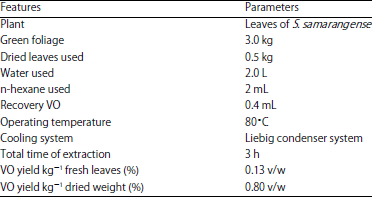
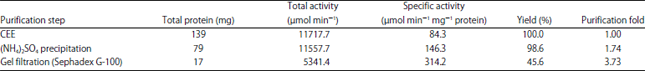
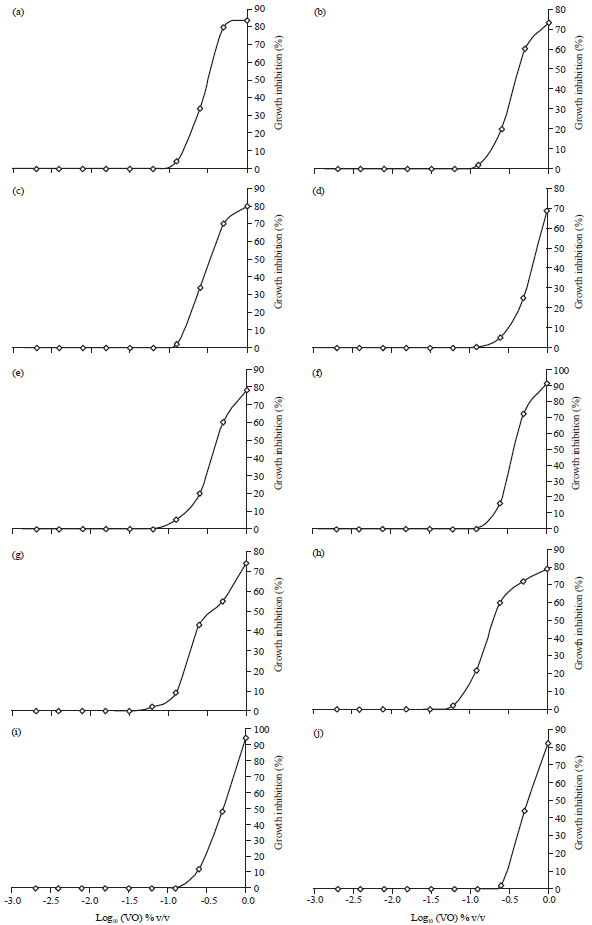
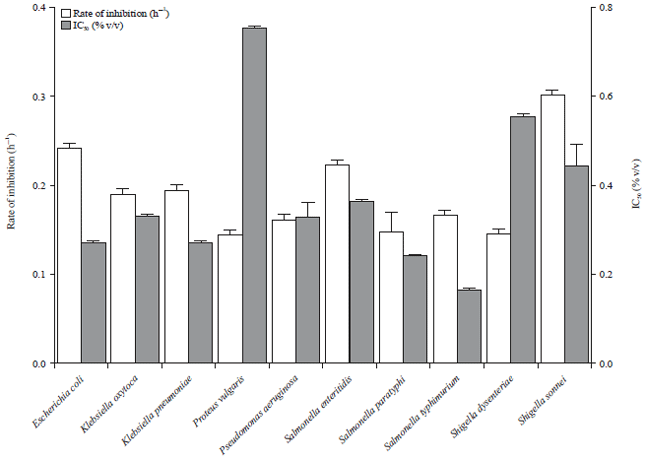
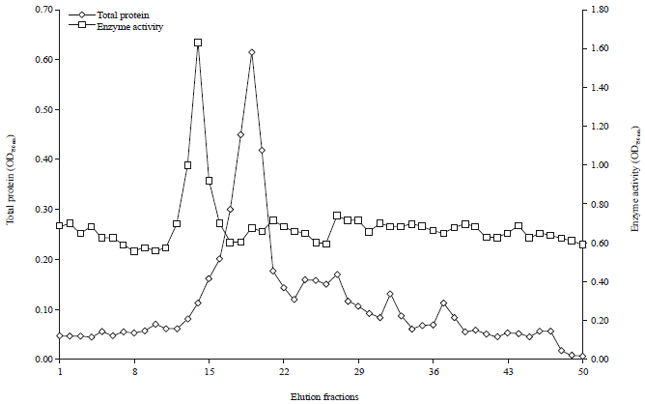
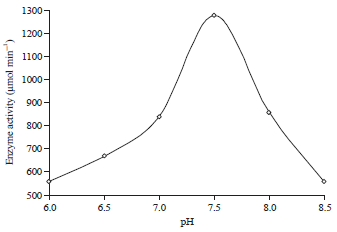
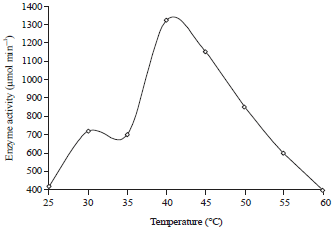
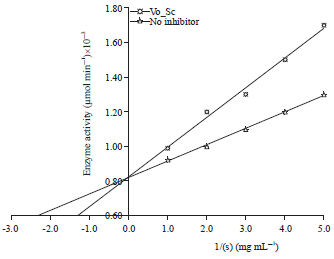
Research Article
Syzygium samarangense Volatile Oil Inhibited Bacteria Growth and Extracellular Protease of Salmonella typhimurium
Department of Microbial, Biochemical and Food Biotechnology, Faculty of Natural and Agricultural Sciences, University of the Free State, 9301 Bloemfontein, South Africa
LiveDNA: 27.30539
S. Adeola
Department of Biochemistry, Faculty of Science, Lagos State University, P.O. Box 0001, Lasu Post Office, Ojo, Lagos, Nigeria
A.O. Giwa Ajeniya
Department of Chemistry, Faculty of Science, Lagos State University, P.O. Box 0001, Lasu Post Office, Ojo, Lagos, Nigeria
LiveDNA: 234.23067