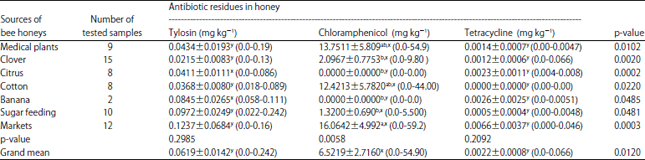
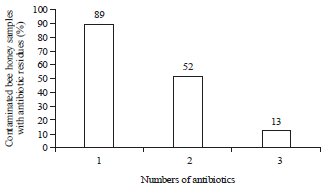
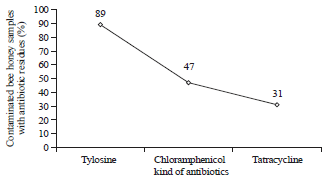
Research Article
Residues of Tetracycline, Chloramphenicol and Tylosin Antibiotics in the Egyptian Bee Honeys Collected from Different Governorates
Department of Economic Entomology and Pesticides, Faculty of Agriculture, Cairo University, 12613 Giza, Egypt
LiveDNA: 20.30729