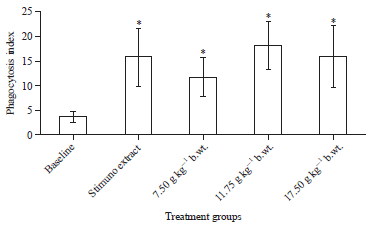
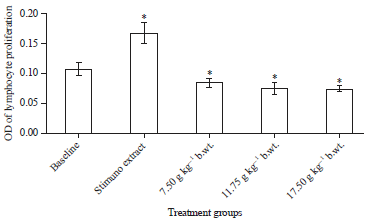
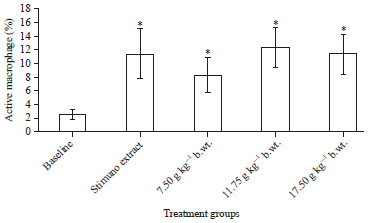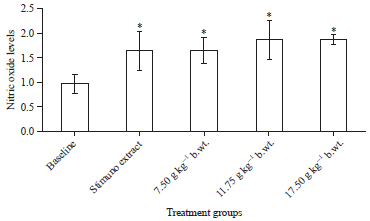Research Article
In vivo Immunomodulatory Activity of Faloak Bark Extract (Sterculia quadrifida R.Br)
Department of Pharmaceutical Biology, Faculty of Medicine and Health Sciences, Universitas Muhammadiyah Yogyakarta, Bantul, Yogyakarta, Indonesia
LiveDNA: 62.28520
Triana Hertiani
Department of Pharmaceutical Biology, Faculty of Pharmacy, Universitas Gadjah Mada, Sekip Utara, Yogyakarta, Indonesia
LiveDNA: 62.23946
Purwantiningsih
Department of Pharmacology and Clinical Pharmacy, Faculty of Pharmacy, Universitas Gadjah Mada, Sekip Utara, Yogyakarta, Indonesia
Siswadi
House of Research Environment and Forestry, Jl. Alfons Nisnoni No. 7B, Airnona, Kupang, Indonesia