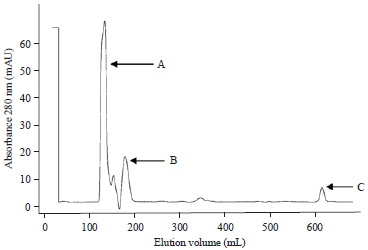
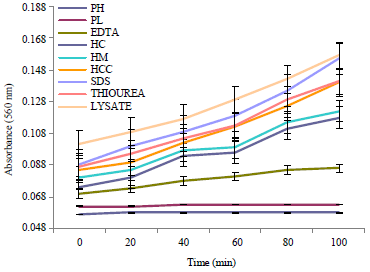
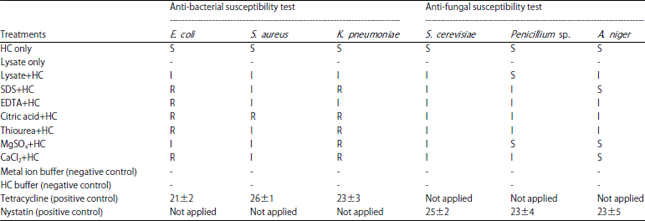
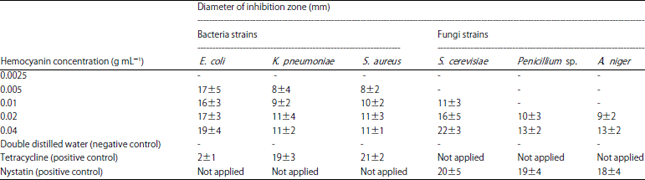
Research Article
Broad-spectrum Antimicrobial Activity of Purified Hemocyanin Subunit IIIA Isolated from Asian Horseshoe Crab, Tachypleus gigas
Horseshoe Crab Research Group, Institute of Marine Biotechnology, Universiti Malaysia Terengganu, 21030 Kuala Nerus, Terengganu, Malaysia
S.K. Dzulkiply
Horseshoe Crab Research Group, Institute of Marine Biotechnology, Universiti Malaysia Terengganu, 21030 Kuala Nerus, Terengganu, Malaysia
M.A. Yusof
Horseshoe Crab Research Group, Institute of Marine Biotechnology, Universiti Malaysia Terengganu, 21030 Kuala Nerus, Terengganu, Malaysia
N.A. Kamaruding
Horseshoe Crab Research Group, Institute of Marine Biotechnology, Universiti Malaysia Terengganu, 21030 Kuala Nerus, Terengganu, Malaysia
N. Ismail
Horseshoe Crab Research Group, Institute of Marine Biotechnology, Universiti Malaysia Terengganu, 21030 Kuala Nerus, Terengganu, Malaysia
LiveDNA: 60.26983